Berikut merupakan kutipan ilmiah Mikrobiologi Kedokteran yang bermanfaat sehingga disusun dan digunakan sebagai referensi pribadi.
Perpustakaan Keluarga : Helmut Todo Tua Simamora dan dr. Olga Y.V Hutapea
A U.S. vector biologist appears to have accidentally written virological history simply by having sex with his wife after returning from a field trip to Senegal. A study just released in Emerging Infectious Diseases suggests that the researcher, Brian Foy of Colorado State University in Fort Collins, passed to his wife the Zika virus, an obscure pathogen that causes joint pains and extreme fatigue. If so, it would be the first documented case of sexual transmission of an insect-borne disease.
Foy is the first author of the paper, which describes three anonymous patients. But in an interview with Science, he confirmed that he is the anonymous "patient 1"; his Ph.D. student Kevin Kobylinski, who accompanied him on the trip to Senegal and also got sick, is "patient 2." Foy's wife, Joy Chilson Foy, a nurse at the Poudre Valley Hospital in Fort Collins, is "patient 3"; she is also a co-author of the paper.
Exactly what happened when Foy and Kobylinski returned from Senegal on 24 August 2008 has remained a mystery for years. As part of their research on malaria, the scientists had been collecting mosquitoes in a southeastern village called Bandafassi, where they were often bitten. About 5 days after their return, both researchers got sick. Both had a rash on their torso, extreme fatigue, headaches, and swollen and painful wrists, knees, and ankles. Foy also had symptoms of prostatitis, including painful urination, and he and his wife noticed what looked like blood in his semen, according to the paper.
On 3 September, Foy's wife's fell ill as well, with malaise, chills, extreme headache, hypersensitivity to light, and muscle pains. The couple's four children remained healthy. The symptoms started receding within about a week in all three patients, although the joint pains lingered.
The scientists suspected they were infected through one of their many mosquito bites but were stumped as to the pathogen. So were several laboratories, including the U.S. Centers for Disease Control and Prevention (CDC), whose lab for insect-borne diseases is in Fort Collins. Antibody tests on serum samples from the two scientists tested positive for dengue, a viral disease that might have explained the symptoms, but samples from Chilson Foy came back negative. "Eventually, the CDC said, 'We think you had dengue, but we don't know what your wife had,' " says Foy, who decided to keep samples from all three in the freezer.
The mystery might never have been solved if Kobylinski hadn't gone out for a few beers with Andrew Haddow, a medical entomologist at the University of Texas Medical Branch (UTMB) at Galveston, during another trip to Senegal more than a year later. Haddow, who studies how pathogens survive in the jungle and emerge when humans encroach, had a great personal interest in Zika: His grandfather, Alexander Haddow, was one of three scientists who had isolated the virus from a rhesus monkey in the Zika Forest near Entebbe, Uganda, in 1947 and described it in a paper in 1952. "I read all of my grandfather's papers, so that stuff really interests me," Haddow says.
Zika, he speculated, might account for the trio's symptoms—even though "it was just a hunch." After Kobylinski returned home in December 2009 and told Foy about the encounter, they decided to send the samples to Haddow, who asked his UTMB colleague Robert Tesh, a veteran virologist, to run a battery of tests, including one for the Zika virus. Sure enough, all three samples had antibodies to the virus. "Then it all fell into place," Foy says. The dengue antibodies were a red herring, he says: both researchers had been vaccinated for the yellow fever virus, which is closely related to dengue; antibodies against these viruses often cross-react.
There is no direct evidence that Foy's wife was infected through sexual contact, but the circumstantial evidence is strong. It's very unlikely that she was infected by a bite by a mosquito that first bit her husband; the three tropical Aedes mosquito species known to transmit Zika don't live in northern Colorado, and moreover, the virus has to complete a 2-week life cycle within the insect before it can infect the next human; Foy's wife fell ill just 9 days after his return. And yes, as the paper puts it, "patients 1 and 3 reported having vaginal sexual intercourse in the days after patient 1 returned home but before the onset of his clinical illness." ("My wife wasn't happy with what happened afterwards," Foy adds.)
Not much is known about the Zika virus, which is found in many parts of Africa and Southeast Asia. Researchers from the Pasteur Institute in Dakar reported in 1993 that it occurs in southeastern Senegal, where Foy and Kobylinski work, but it's unknown how often it causes disease. Most cases are never reported or are mistaken for dengue, a much more widespread disease, Haddow says. In fact, only 14 Zika cases had ever been described in the medical literature until an explosive outbreak occurred in 2007 on Yap, an island in the Federated States of Micronesia, where it had never been seen before. An extensive investigation of that epidemic by CDC concluded that 73% of the population was infected, an impressive number that suddenly made Zika an emerging pathogen to watch.
Foy and his co-authors believe sexual transmission of a mosquito-borne virus has never been reported before, although there were hints from the literature that it might be possible. Boars experimentally infected with the Japanese encephalitis virus, for instance, shed the virus in their semen, and female pigs artificially inseminated with it become infected.
Scientists already knew that many insect-borne pathogens can be transmitted orally as well, says medical entomologist Paul Reiter of the Pasteur Institute in Paris. So even if it hasn't been documented before, it's not a big surprise that infected semen deposited inside the vagina could cause an infection, he says.
What's still unclear is how important sexual transmission is in Zika's epidemiology. Haddow believes it plays a very minor role at best and that the vast majority of cases occur through mosquito bites. Yet a few data points from the Yap outbreak hint that sexual transmission may have played a role, Foy says. The population aged between 30 and 59 was hardest hit, and among women, the so-called attack rate—the percentage of people who get sick—was almost 50% higher than among men. (With most sexually transmitted infections, vaginal intercourse poses a higher risk of infection for women.) But there could be other explanations for that as well; Foy says he's interested in studying the issue.
Haddow is doing more research on Zika as well. Although he never really knew his grandfather, he says that stories about his career inspired him to become a virus hunter, and helping solve the Fort Collins riddle, he adds, was "a very good feeling. ... I think my grandfather would have been happy too."
Flaviviridae
VIRION
A Viral Biorealm page on the family Flaviviridae
Baltimore Classification
Higher order taxa
Viruses; ssRNA positive-strand viruses, no DNA stage; Flaviviridae
Genera
Flavivirus, Pestivirus, Hepacivirus
Description and Significance
Flaviviridae contains a myriad of viruses that cause disease in humans. Flaviviridae has a total of 69 pathogens in its rank. The family gets its name from Yellow Fever virus, a type virus of Flaviviridae; flavus means yellow in Latin.
The Flavivirus genus contains several nasty critters that include yellow fever virus, dengue fever virus, and Japanese encephalitis (JE) virus. The pestivirus genus has three serotypes of bovine viral diarrhea, but no human pathogens are yet known. Genus Hepacivirus consists of hepatitis C virus and its relatives. (source: Stanford.edu)
Genome Structure
The genome of Flaviviridae is not segmented and contains a single molecule of linear positive-sense, single-stranded RNA. The complete genome is 9500-12500 nucleotides long. The 5'-end of the genome has a methylated nucleotide cap, or genome-linked protein (VPg). The 3'- terminus has no poly (A) tract but the poly (A) tract is present in some strains of tick-born encephalitis complex of flaviviruses. (source: ICTV dB Descriptions)
Virion Structure of a Flaviviridae
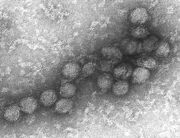
The virions of flaviviridae consist of an envelope and a nucleocapsid. The virus capsid is enveloped. The virions ar spherical to pleomorphic and measure 40-60 nm in diameter. There are small spikes surrounded by a prominent fringe that make up the surface projections. The capsid is round and exhibits a polyhedral symmetry. The core is isometric and has a diameter of 25-30 nm. The capsids can be penetrated by stain and some appear dark in the center. (source: ICTV dB Descriptions)
Reproduction Cycle of a Flaviviridae in a Host Cell
Viral Ecology & Pathology
Many species of Flaviviridae can replicate in both mammalian and insect cells. They can survive for long periods in hosts such as ticks by replicating in this host without damage to the insect.
Flaviviridae viruses produce a wide range of diseases including fever, arthralgia, rash, haemorrhagic fever and encephalitis. The outcome of infection is influenced by both the virus and a number of host-specific factors like age, sex, genetic susceptibility and preexposure to same or related agent.
| LinkOut | Subject | LinkOut Provider |
| Flaviviridae | taxonomy/phylogenetic | Encyclopedia of life |
| Flaviviridae | taxonomy/phylogenetic | International Committee on Taxonomy of Viruses |
| Flaviviridae | taxonomy/phylogenetic | World Register of Marine Species |
| Wikipedia | taxonomy/phylogenetic | iPhylo |

Enveloped, spherical, about 50 nm in diameter. The surface proteins are arranged in an icosahedral-like symmetry.
Source: Zhang et al
Source: Zhang et al
GENOME

Monopartite, linear, ssRNA(+) genome of about 9.7-12 kb. The genome 3’ terminus is not polyadenylated but forms a loop structure. The 5’ end has a methylated nucleotide cap (to allow translation) or a genome-linked protein (VPg).
GENE EXPRESSION
The virion RNA is infectious and serves as both the genome and the viral messenger RNA. The whole genome is translated into a polyprotein, which is processed co- and post-translationally by host and viral proteases.
REPLICATION
CYTOPLASMIC
- Attachement of the viral envelope protein E to host receptors mediates internalization into the host cell by clathrin-mediated endocytosis or by apoptotic mimicry
- Fusion of virus membrane with host endosomal membrane. RNA genome is released into the cytoplasm.
- The positive-sense genomic ssRNA is translated into a polyprotein, which is cleaved into all structural and non structural proteins (to yield the replication proteins).
- Replication takes place at the surface of endoplasmic reticulum in cytoplasmic viral factories. A dsRNA genome is synthesized from the genomic ssRNA(+).
- The dsRNA genome is transcribed/replicated thereby providing viral mRNAs/new ssRNA(+) genomes.
- Virus assembly occurs at the endoplasmic reticulum and seems to be facilitated by theviral ionic channel p7. The virion buds via the host ESCRT complexes at the endoplasmic reticulum, is transported to the Golgi apparatus.
- Release of new virions by exocytosis.
Flaviviridae is a family of viruses. Humans and other mammals serve as natural hosts. They are primarily spread through arthropod vectors (mainly ticks and mosquitoes). The family gets its name from the Yellow Fever virus, the type virus ofFlaviviridae; flavus means yellow in Latin. (Yellow fever in turn was named because of its propensity to cause jaundice in victims.)[1] There are currently over 100 species in this family, divided among four genera. Diseases associated with this family include: hepaciviruses: hepatitis; pestiviruses: hemorrhagic syndromes, abortion, fatal mucosal disease; flavivirus: hemorrhagic fever, encephalitis.[2][3]
| Flaviviridae | |
|---|---|
| Virus classification | |
| Group: | Group IV ((+)ssRNA) |
| Family: | Flaviviridae |
| Genera | |
Taxonomy
- Genus Flavivirus (type species Yellow fever virus, others include West Nile virus, Dengue Fever and Zika virus)—contains 67 identified human and animal viruses
- Genus Hepacivirus (type species Hepatitis C virus,[4] also includes GB virus B)
- Genus Pegivirus (includes GB virus A, GB virus C, and GB virus D)
- Genus Pestivirus (type species bovine viral diarrhea virus, others include classical swine fever or hog cholera)—contains viruses infecting non-human mammals
Group: ssRNA(+)
Tidak ada komentar:
Posting Komentar