Berikut merupakan kutipan ilmiah yang bermanfaat sehingga disusun dan digunakan sebagai referensi pribadi.
A cyanide is any chemical compound that contains monovalent combining group CN. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom.[1]
In inorganic cyanides, such as sodium cyanide, NaCN, this group is present as the negatively charged polyatomiccyanide ion (CN−); these compounds, which are regarded as salts of hydrocyanic acid, are highly toxic. The cyanide ion is isoelectronic with carbon monoxide and with molecular nitrogen.[2][3] Most cyanides are highly toxic.[4]
Organic cyanides are usually called nitriles; in these, the CN group is linked by a covalent bond to a carbon-containing group, such as methyl (CH3) in methyl cyanide (acetonitrile).
Hydrocyanic acid, also known as hydrogen cyanide, or HCN, is a highly volatile liquid used to prepare acrylonitrile, which is used in the production of acrylic fibers, synthetic rubber, and plastics. Cyanides are employed in a number of chemical processes, including fumigation, case hardening of iron and steel, electroplating, and the concentration ofores. In nature, substances yielding cyanide are present in certain seeds, such as the pit of the cherry and the seeds ofapples.
Cyanide toxicity is generally considered to be a rare form of poisoning. However, cyanide exposure occurs relatively frequently in patients with smoke inhalation from residential or industrial fires. In addition, intensive treatment with sodium nitroprusside or long-term consumption of cyanide-containing foods is a possible source of cyanide poisoning. Historically, cyanide has been used as a chemical warfare agent, and it could potentially be an agent for a terrorist attack.[1, 2]
Cyanide exists in gaseous, liquid, and solid forms. Hydrogen cyanide (HCN, also known as prussic acid) is a volatile liquid that boils at 25.6° C (78.1° F). Potassium and sodium cyanide salts are water soluble, whereas mercury, copper, gold, and silver cyanide salts are poorly water soluble.
In addition, a number of cyanide-containing compounds, known as cyanogens, may release cyanide during metabolism. These include, but are not limited to, cyanogen chloride and cyanogen bromide (gases with potent pulmonary irritant effects), nitriles (R-CN), and the vasodilator nitroprusside sodium, which may produce iatrogenic cyanide poisoning during prolonged or high-dose intravenous (IV) therapy (>10 mcg/kg/min). (See Etiology.)
Industry widely uses nitriles as solvents and in the manufacturing of plastics. Nitriles may release HCN during burning or when metabolized after absorption by the skin or gastrointestinal tract. A number of synthesized and natural compounds produce HCN when burned. These combustion gases likely contribute to the morbidity and mortality from smoke inhalation. Finally, long-term consumption of cyanide-containing foods, such as cassava root or apricot seeds,[3] may lead to cyanide poisoning.
Depending on its form, cyanide may cause toxicity through inhalation, ingestion, dermal absorption, or parenteral administration. Clinical manifestations vary widely, depending on the dose and route of exposure, and may range from minor upper airway irritation to cardiovascular collapse and death within minutes. (See Clinical Presentation.) In severe cases, rapid, aggressive therapy consisting of supportive care and antidote administration can be lifesaving. (See Treatment andMedication.)
Cyanide as a chemical weapon
HCN (North Atlantic Treaty Organization [NATO] designation AC) is one of two cyanide chemical warfare agents[4, 5, 6] ; the other is cyanogen chloride (NATO designation CK). Cyanide is a rapidly lethal agent when used in enclosed spaces where high concentrations can be achieved easily.[7, 8, 9, 10] In addition, because of the extensive use of cyanide in industry in the United States, this agent presents a credible threat for terrorist use.[5]
Cyanide was first used as a chemical weapon in the form of gaseous HCN in World War I. Starting in 1915, the French military used approximately 4000 tons of cyanide, without notable success. The failure of this measure was probably attributable to the high volatility of cyanide and the inability of the 1- to 2-lb munitions used to deliver the amounts of chemical required for biologic effects.[5, 6]
The introduction of cyanogen chloride by the French in 1916 made available a compound that, being both more toxic and less volatile, was a more effective chemical agent. Other alleged military uses of cyanide include Japanese attacks on China before and during World War II and Iraqi attacks on Kurds in the 1980s.
 | |
| Names | |
|---|---|
| Systematic IUPAC name
Cyanide
| |
| Identifiers | |
| ChEBI | CHEBI:17514 |
| ChemSpider | 5755 |
| Jmol interactive 3D | Image |
| PubChem | 5975 |
| Properties | |
| CN− | |
| Molar mass | 26.02 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
| Infobox references | |

In IUPAC nomenclature, organic compounds that have a –C≡N functional group are called nitriles. Thus, nitriles are organic compounds.[5][6] An example of a nitrile is CH3CN, acetonitrile, also known as methyl cyanide. Nitriles usually do not release cyanide ions. A functional group with a hydroxyl and cyanide bonded to the same carbon is called cyanohydrin. Unlike nitriles, cyanohydridins do release hydrogen cyanide. In inorganic chemistry, salts containing the C≡N− ion are referred to as cyanides.
The word is derived from the Greek kyanos, meaning dark blue, as a result of its being first obtained by the heating of the pigment known as Prussian blue.
In nature
Cyanides are produced by certain bacteria, fungi, and algae and are found in a number of plants. Cyanides are found in substantial amounts in certain seeds and fruit stones, e.g., those of apricots, apples, and peaches.[7] In plants, cyanides are usually bound tosugar molecules in the form of cyanogenic glycosides and defend the plant against herbivores. Cassava roots (also called manioc), an important potato-like food grown in tropical countries (and the base from which tapioca is made), also contain cyanogenic glycosides.[8][9]
The Madagascar bamboo Cathariostachys madagascariensis produces cyanide as a deterrent to grazing. In response, the golden bamboo lemur, which eats the bamboo, has developed a high tolerance to cyanide.
Interstellar medium
The cyanide radical CN· has been identified in interstellar space.[10] The cyanide radical (called cyanogen) is used to measure the temperature of interstellar gas clouds.[11]
Pyrolysis and combustion product
Hydrogen cyanide is produced by the combustion or pyrolysis of certain materials under oxygen-deficient conditions. For example, it can be detected in the exhaustof internal combustion engines and tobacco smoke. Certain plastics, especially those derived from acrylonitrile, release hydrogen cyanide when heated or burnt.[12]
Coordination chemistry
The cyanide anion is a ligand for many transition metals.[13] The high affinities of metals for this anion can be attributed to its negative charge, compactness, and ability to engage in π-bonding. Well-known complexes include:
- hexacyanides [M(CN)6]3− (M = Ti, V, Cr, Mn, Fe, Co), which are octahedral in shape;
- the tetracyanides, [M(CN)4]2− (M = Ni, Pd, Pt), which are square planar in their geometry;
- the dicyanides [M(CN)2]− (M = Cu, Ag, Au), which are linear in geometry.
The dye Prussian blue was first accidentally made around 1706, by heating substances containing iron and carbon and nitrogen. Prussian blue consists of an iron-containing compound called "ferrocyanide" ({Fe(CN)6]4−) meaning "blue substance with iron", from Latin ferrum = "iron" and Greek kyanos = "(dark) blue".[14]Prussian blue is the deep-blue pigment used in the making of blueprints.
The enzymes called hydrogenases contain cyanide ligands attached to iron in their active sites. The biosynthesis of cyanide in the [NiFe]-hydrogenases proceeds from carbamoyl phosphate, which converts to cysteinyl thiocyanate, the CN− donor.[15]
Organic derivatives
Main article: Nitriles
Because of the cyanide anion's high nucleophilicity, cyano groups are readily introduced into organic molecules by displacement of a halide group (e.g., the chlorideon methyl chloride). In general, organic cyanides are called nitriles. Thus, CH3CN can be called methyl cyanide but more commonly is referred to as acetonitrile. In organic synthesis, cyanide is a C-1 synthon; i.e., it can be used to lengthen a carbon chain by one, while retaining the ability to be functionalized.[citation needed]
- RX + CN− → RCN + X− (nucleophilic substitution) followed by
- RCN + 2 H2O → RCOOH + NH3 (hydrolysis under reflux with mineral acid catalyst), or
- 2 RCN + LiAlH4 + (second step) 4 H2O → 2 RCH2NH2 + LiAl(OH)4 (under reflux in dry ether, followed by addition of H2O)
Manufacture
Main article: Hydrogen cyanide § Production and synthesis
The principal process used to manufacture cyanides is the Andrussow process in which gaseous hydrogen cyanide is produced from methane and ammonia in the presence of oxygen and a platinum catalyst.[16][17]
- 2 CH4 + 2 NH3 + 3 O2 → 2 HCN + 6 H2O
Gaseous hydrogen cyanide may be dissolved in aqueous sodium hydroxide solution to produce sodium cyanide.[citation needed]
Toxicity
Main article: Cyanide poisoning
Many cyanides are highly toxic. The cyanide anion is an inhibitor of the enzyme cytochrome c oxidase (also known as aa3) in the fourth complex of the electron transport chain (found in the membrane of the mitochondria of eukaryotic cells). It attaches to the iron within this protein. The binding of cyanide to this enzyme prevents transport of electrons from cytochrome c to oxygen. As a result, the electron transport chain is disrupted, meaning that the cell can no longer aerobically produce ATP for energy.[18] Tissues that depend highly on aerobic respiration, such as the central nervous system and the heart, are particularly affected. This is an example of histotoxic hypoxia.[19]
The most hazardous compound is hydrogen cyanide, which is a gas at ambient temperatures and pressure and can therefore be inhaled. For this reason, an air respirator supplied by an external oxygen source must be worn when working with hydrogen cyanide. Hydrogen cyanide is produced when a solution containing alabile cyanide is made acidic, because HCN is a weak acid. Alkaline solutions are safer to use because they do not evolve hydrogen cyanide gas. Hydrogen cyanide may be produced in the combustion of polyurethanes; for this reason, polyurethanes are not recommended for use in domestic and aircraft furniture. Oral ingestion of a small quantity of solid cyanide or a cyanide solution as little as 200 mg, or to airborne cyanide of 270 ppm is sufficient enough to cause death within minutes.[19]
Organic nitriles do not readily release cyanide ions, and so have low toxicities. By contrast, compounds such as trimethylsilyl cyanide (CH3)3SiCN readily release HCN or the cyanide ion upon contact with water.[citation needed]
Antidote
Hydroxocobalamin reacts with cyanide to form cyanocobalamin, which can be safely eliminated by the kidneys. This method has the advantage of avoiding the formation of methemoglobin (see below). This antidote kit is sold under the brand name Cyanokit and was approved by the FDA in 2006.[20]
An older cyanide antidote kit included administration of three substances: amyl nitrite pearls (administered by inhalation), sodium nitrite, and sodium thiosulfate. The goal of the antidote was to generate a large pool of ferric iron (Fe3+) to compete for cyanide with cytochrome a3 (so that cyanide will bind to the antidote rather than the enzyme). The nitrites oxidize hemoglobin to methemoglobin, which competes with cytochrome oxidase for the cyanide ion. Cyanmethemoglobin is formed and the cytochrome oxidase enzyme is restored. The major mechanism to remove the cyanide from the body is by enzymatic conversion to thiocyanate by themitochondrial enzyme rhodanese. Thiocyanate is a relatively non-toxic molecule and is excreted by the kidneys. To accelerate this detoxification, sodium thiosulfate is administered to provide a sulfur donor for rhodanese, needed in order to produce thiocyanate.[21]
Sensitivity
Minimum risk levels (MRLs) may not protect for delayed health effects or health effects acquired following repeated sublethal exposure, such as hypersensitivity,asthma, or bronchitis. MRLs may be revised after sufficient data accumulates.[22]
Applications
Mining
Main article: Gold cyanidation
Cyanide is mainly produced for the mining of gold and silver: It helps dissolve these metals and their ores. In the cyanide process, finely ground high-grade ore is mixed with the cyanide (concentration of about two kilogram NaCN per tonne); low-grade ores are stacked into heaps and sprayed with a cyanide solution (concentration of about one kilogram NaCN per ton). The precious metals are complexed by the cyanide anions to form soluble derivatives, e.g., [Au(CN)2]− and [Ag(CN)2]−.[23]
-
- 4 Au + 8 NaCN + O2 + 2 H2O → 4 Na[Au(CN)2] + 4 NaOH
Silver is less "noble" than gold and often occurs as the sulfide, in which case redox is not invoked (no O2 is required). Instead, a displacement reaction occurs:
-
- Ag2S + 4 NaCN + H2O → 2 Na[Ag(CN)2] + NaSH + NaOH
The "pregnant liquor" containing these ions is separated from the solids, which are discarded to a tailing pond or spent heap, the recoverable gold having been removed. The metal is recovered from the "pregnant solution" by reduction with zinc dust or by adsorption onto activated carbon. This process can result in environmental and health problems. A number of environmental disasters have followed the overflow of tailing ponds at gold mines. Cyanide contamination of waterways has resulting in numerous cases of human and aquatic species mortality.[citation needed]
Aqueous cyanide is hydrolyzed rapidly, especially in sunlight. It can mobilize some heavy metals such as mercury if present. Gold can also be associated with arsenopyrite (FeAsS), which is similar to iron pyrite (fool's gold), wherein half of the sulfur atoms are replaced by arsenic. Gold-containing arsenopyrite ores are similarly reactive toward inorganic cyanide.[citation needed]
Cyanide is also used in electroplating, where it stabilizes metal ions in the electrolyte solution prior to their deposition.[citation needed]
Industrial organic chemistry
Some nitriles are produced on a large scale, e.g., adiponitrile is a precursor to nylon. Such compounds are often generated by combining hydrogen cyanide and alkenes, i.e., hydrocyanation: RCH=CH2 + HCN → RCH(CN)CH3. Metal catalysts are required for such reactions.
Medical uses
The cyanide compound sodium nitroprusside is used mainly in clinical chemistry to measure urine ketone bodies mainly as a follow-up to diabetic patients. On occasion, it is used in emergency medical situations to produce a rapid decrease in blood pressure in humans; it is also used as a vasodilator in vascular research. The cobalt in artificial vitamin B12 contains a cyanide ligand as an artifact of the purification process; this must be removed by the body before the vitamin molecule can be activated for biochemical use. During World War I, a copper cyanide compound was briefly used by Japanese physicians for the treatment of tuberculosisand leprosy.[24]
Illegal fishing and poaching
Main article: Cyanide fishing
Cyanides are illegally used to capture live fish near coral reefs for the aquarium and seafood markets. The practice is controversial, dangerous, and damaging but is driven by the lucrative exotic fish market.[citation needed]
Poachers in Africa have been known to use cyanide to poison waterholes, to kill elephants for their ivory.[25]
Pest control
Cyanide is used for pest control in New Zealand particularly for possums, an introduced marsupial that threatens the conservation of native species and spreadstuberculosis amongst cattle. Possums can become bait shy but the use of pellets containing the cyanide reduces bait shyness. Cyanide has been known to kill native birds, including the endangered kiwi.[26] Cyanide is also effective for controlling the dama wallaby, another introduced marsupial pest in New Zealand.[27] A licence is required to store, handle and use cyanide in New Zealand.
Niche uses
Potassium ferrocyanide is used to achieve a blue color on cast bronze sculptures during the final finishing stage of the sculpture. On its own, it will produce a very dark shade of blue and is often mixed with other chemicals to achieve the desired tint and hue. It is applied using a torch and paint brush while wearing the standard safety equipment used for any patina application: rubber gloves, safety glasses, and a respirator. The actual amount of cyanide in the mixture varies according to the recipes used by each foundry.
Cyanide is also used in jewelry-making and certain kinds of photography such as sepia toning.
Cyanides are used as insecticides for fumigating ships. Cyanide salts are used for killing ants, and have in some places been used as rat poison (the less toxic poison arsenic is more common[citation needed]).
Although usually thought to be toxic, cyanide and cyanohydrins have been demonstrated to increase germination in various plant species.[28][29]
Human poisoning
Deliberate cyanide poisoning of humans has occurred many times throughout history.[30] For notable cyanide deaths, see Cyanide poisoning: Historical cases.
Most significantly, hydrogen cyanide released from pellets of Zyklon-B was used extensively in the systematic mass murders of the Holocaust, especially in extermination camps. Poisoning by hydrogen cyanide gas within a gas chamber (as a salt of hydrocyanic acid is dropped into a strong acid, usually sulfuric acid) is one method of executing a condemned prisoner as the condemned prisoner eventually breathes the lethal fumes.
Food additive
Due to the high stability of their complexation with iron, ferrocyanides (Sodium ferrocyanide E535, Potassium ferrocyanide E536, and Calcium ferrocyanide E538[31]) do not decompose to lethal levels in the human body and are used in the food industry as, e.g., an anticaking agent in table salt.[32]
Chemical tests for cyanide
Prussian blue
Iron(II) sulfate is added to a solution suspected of containing cyanide, such as the filtrate from the sodium fusion test. The resulting mixture is acidified with mineral acid. The formation of Prussian blue is a positive result for cyanide.
para-Benzoquinone in DMSO
A solution of para-benzoquinone in DMSO reacts with inorganic cyanide to form a cyanophenol, which is fluorescent. Illumination with a UV light gives a green/blue glow if the test is positive.[33]
Copper and an aromatic amine
As used by fumigators to detect hydrogen cyanide, copper(II) salt and an aromatic amine such as benzidine is added to the sample; as an alternative to benzidine an alternative amine di-(4,4-bis-dimethylaminophenyl) methane can be used. A positive test gives a blue color. Copper(I) cyanide is poorly soluble. By sequestering the copper(I) the copper(II) is rendered a stronger oxidant. The copper, in a cyanide facilitated oxidation, converts the amine into a colored compound. The Nernst equation explains this process. Another good example of such chemistry is the way in which the saturated calomel reference electrode (SCE) works. The copper, in a cyanide-facilitated oxidation, converts the amine into a colored compound.
Pyridine-barbituric acid colorimetry
A sample containing inorganic cyanide is purged with air from a boiling acid solution into a basic absorber solution. The cyanide salt absorbed in the basic solution is buffered at pH 4.5 and then reacted with chlorine to form cyanogen chloride. The cyanogen chloride formed couples pyridine with barbituric acid to form a strongly colored red dye that is proportional to the cyanide concentration. This colorimetric method following distillation is the basis for most regulatory methods (for instance EPA 335.4) used to analyze cyanide in water, wastewater, and contaminated soils. Distillation followed by colorimetric methods, however, have been found to be prone to interferences from thiocyanate, nitrate, thiosulfate, sulfite, and sulfide that can result in both positive and negative bias. It has been recommended by the USEPA (MUR March 12, 2007) that samples containing these compounds be analyzed by Gas-Diffusion Flow Injection Analysis — Amperometry.[citation needed]
Gas diffusion flow injection analysis — amperometry
Instead of distilling, the sample is injected into an acidic stream where the HCN formed is passed under a hydrophobic gas diffusion membrane that selectively allows only HCN to pass through. The HCN that passes through the membrane is absorbed into a basic carrier solution that transports the CN to an amperometric detector that accurately measures cyanide concentration with high sensitivity. Sample pretreatment determined by acid reagents, ligands, or preliminary UV irradiation allow cyanide speciation of free cyanide, available cyanide, and total cyanide respectively. The relative simplicity of these flow injection analysis methods limit the interference experienced by the high heat of distillation and also prove to be cost effective since time consuming distillations are not required.
Administer a cyanide antidote if the diagnosis of cyanide toxicity is strongly suspected, without waiting for laboratory confirmation. Available antidotes are hydroxocobalamin (Cyanokit) and sodium thiosulfate and sodium nitrite (Nithiodote). Both are given intravenously.
Patients who present with more than minimal symptoms that resolve without treatment should be admitted for observation and supportive care. In patients with acute poisoning from hydrogen cyanide (HCN) gas or soluble salts, the principal acute care concerns are hemodynamic instability and cerebral edema. The continuous cardiac monitoring, respiratory and cardiovascular support, and frequent neurologic evaluation these patients require is generally best provided in an intensive care unit.
Conversely, acute poisoning from cyanogens (nitriles) or poorly soluble salts may not manifest or become life-threatening for several hours after exposure. These patients require a 24-hour observation period.[23, 24]
Oxygenation should be optimized and continuous cardiac monitoring provided. Depending on the severity of symptoms, endotracheal intubation may be necessary to optimize oxygen delivery and protect the airway. Serum lactate concentrations, chemistries, and arterial or venous blood gases should be monitored.
Patients should be reevaluated 7-10 days after discharge from the hospital.[25]Delayed onset of Parkinson-like syndrome or neuropsychiatric sequelae may be noted on followup.
Special concerns in pregnancy
Fetal demise is possible in cyanide poisoning. Aggressive support and antidotal treatment of the mother is paramount. An obstetric evaluation following stabilization of the mother is essential. Therapeutic abortion may be indicated in the presence of fetal demise.
Cyanide exposure most often occurs via inhalation or ingestion, but liquid cyanide can be absorbed through the skin or eyes. Once absorbed, cyanide enters the blood stream and is distributed rapidly to all organs and tissues in the body.[13]
Inside cells, cyanide attaches itself to ubiquitous metalloenzymes, rendering them inactive. Its principal toxicity results from inactivation of cytochrome oxidase (at cytochrome a3), thus uncoupling mitochondrial oxidative phosphorylation and inhibiting cellular respiration, even in the presence of adequate oxygen stores. Cellular metabolism shifts from aerobic to anaerobic, with the consequent production of lactic acid. Consequently, the tissues with the highest oxygen requirements (brain and heart) are the most profoundly affected by acute cyanide poisoning.
The LCt50 (the concentration-time product capable of killing 50% of the exposed group) for hydrogen cyanide is 2500-5000 mg/min/m3. Vapor exposures in high concentrations (at or above the LCt50) typically can cause death in 6-8 minutes.[1]The lethal oral doses of HCN and cyanide salts are estimated to be 50 mg and 100-200 mg, respectively. For skin exposures, the LD50 (the dose capable of killing 50% of the exposed group) is estimated to be 100 mg/kg.
Cyanogen chloride is used in mining and metalworking, and thus may be involved in an industrial accident. By nature of its chlorine moiety, cyanogen chloride causes irritation of the eyes and respiratory tract and potential delayed pulmonary toxicity similar to chlorine or phosgene gases. In high concentrations (eg, in enclosed spaces), this agent is rapidly acting and lethal, causing death within 6-8 minutes if inhaled at doses at or above its LCt50 of 11,000 mg/min/m3.
Defective cyanide metabolism due to rhodanese deficiency may explain development of Leber optic atrophy, leading to subacute blindness. Cyanide also may cause some of the adverse effects associated with chronic smoking, such as tobacco amblyopia.
Etiology
Smoke inhalation, suicidal ingestion, and industrial exposures are the most frequent sources of cyanide poisoning. Treatment with sodium nitroprusside or long-term consumption of cyanide-containing foods is a possible source. Historically, cyanide has been used as a chemical warfare agent, and it could potentially be an agent for a terrorist attack.[1, 2]
Smoke inhalation
Smoke inhalation during house or industrial fires is the major source of cyanide poisoning in the United States. Individuals with smoke inhalation from enclosed space fires who have soot in the mouth or nose, altered mental status, or hypotension may have significant cyanide poisoning (blood cyanide concentrations >40 mmol/L or approximately 1 mg/L).
Many compounds containing nitrogen and carbon may produce hydrogen cyanide (HCN) gas when burned. Some natural compounds (eg, wool, silk) produce HCN as a combustion product.[4, 14] Household plastics (eg, melamine in dishware, acrylonitrile in plastic cups), polyurethane foam in furniture cushions, and many other synthetic compounds may produce lethal concentrations of cyanide when burned under appropriate conditions of oxygen concentration and temperature.
Intentional poisoning
Cyanide ingestion is an uncommon, but effective, means of suicide.[15] These cases typically involve health-care and laboratory workers who have access to the cyanide salts found in hospital and research laboratories.
Industrial exposure
Countless industrial sources of cyanides exist. Cyanides are used particularly in the metal trades, mining, jewelry manufacturing, dyeing, photography, and agriculture. Specific industrial processes involving cyanide include metal cleaning, reclaiming, or hardening; fumigation; electroplating; and photo processing.[2] In addition, industry uses cyanides in the manufacture of plastics, as reactive intermediates in chemical synthesis, and as solvents (in the form of nitriles).
Exposure to salts and cyanogens occasionally causes poisonings; however, a significant risk for multiple casualties occurs when these products come into contact with mineral acids because HCN gas is produced. A mass casualty incident may develop in an industrial accident in which cyanogen chloride comes in contact with water (eg, during fire-fighting). Containers of cyanogen chloride may rupture or explode if exposed to high heat or following prolonged storage.
Iatrogenic exposure
The vasodilator nitroprusside sodium, when used in high doses or over a period of days, can produce toxic blood concentrations of cyanide. Patients with low thiosulfate reserves (eg, malnourished, postoperative) are at increased risk for developing symptoms, even with therapeutic dosing. Resultant confusion and combativeness initially may be mistaken as intensive care unit (ICU) syndrome (ie, sundowning). Problems may be avoided by coadministration of hydroxocobalamin or sodium thiosulfate.
Ingestion of cyanide-containing supplements or plants
Ingestion of cyanide-containing supplements is rare. Amygdalin (synthetic laetrile, also marketed as vitamin B-17), which contains cyanide, was postulated to have anticancer properties due to the action of cyanide on cancer cells. However, laetrile showed no anticancer activity in human clinical trials in the 1980s and is not available in the United States,[16] although it can be purchased on the Internet.
Amygdalin can be found in the pits of many fruits, such as apricots and papayas; in raw nuts; and in plants such as lima beans, clover, and sorghum. Amygdalin can be hydrolyzed to hydrogen cyanide, and ingestion of large quantities of such foods may result in toxicity.[2]
Human Health Effects
Cyanide is produced in the human body and exhaled in extremely low concentrations with each breath. It is also produced by over 1,000 plant species including sorghum, bamboo and cassava. Relatively low concentrations of cyanide can be highly toxic to people and wildlife.
Cyanide is acutely toxic to humans. Liquid or gaseous hydrogen cyanide and alkali salts of cyanide can enter the body through inhalation, ingestion or absorption through the eyes and skin. The rate of skin absorption is enhanced when the skin is cut, abraded or moist; inhaled salts of cyanide are readily dissolved and absorbed upon contact with moist mucous membranes.
The toxicity of hydrogen cyanide to humans is dependent on the nature of the exposure. Due to the variability of dose-response effects between individuals, the toxicity of a substance is typically expressed as the concentration or dose that is lethal to 50% of the exposed population (LC50 or LD50). The LC50 for gaseous hydrogen cyanide is 100-300 parts per million. Inhalation of cyanide in this range results in death within 10-60 minutes, with death coming more quickly as the concentration increases. Inhalation of 2,000 parts per million hydrogen cyanide causes death within one minute. The LD50 for ingestion is 50-200 milligrams, or 1-3 milligrams per kilogram of body weight, calculated as hydrogen cyanide. For contact with unabraded skin, the LD50 is 100 milligrams (as hydrogen cyanide) per kilogram of body weight.
Although the time, dose and manner of exposure may differ, the biochemical action of cyanide is the same upon entering the body. Once in the bloodstream, cyanide forms a stable complex with a form of cytochrome oxidase, an enzyme that promotes the transfer of electrons in the mitochondria of cells during the synthesis of ATP. Without proper cytochrome oxidase function, cells cannot utilize the oxygen present in the bloodstream, resulting in cytotoxic hypoxia or cellular asphyxiation. The lack of available oxygen causes a shift from aerobic to anaerobic metabolism, leading to the accumulation of lactate in the blood. The combined effect of the hypoxia and lactate acidosis is depression of the central nervous system that can result in respiratory arrest and death. At higher lethal concentrations, cyanide poisoning also affects other organs and systems in the body, including the heart.
Initial symptoms of cyanide poisoning can occur from exposure to 20 to 40 ppm of gaseous hydrogen cyanide, and may include headache, drowsiness, vertigo, weak and rapid pulse, deep and rapid breathing, a bright-red color in the face, nausea and vomiting. Convulsions, dilated pupils, clammy skin, a weaker and more rapid pulse and slower, shallower breathing can follow these symptoms. Finally, the heartbeat becomes slow and irregular, body temperature falls, the lips, face and extremities take on a blue color, the individual falls into a coma, and death occurs. These symptoms can occur from sublethal exposure to cyanide, but will diminish as the body detoxifies the poison and excretes it primarily as thiocyanate and 2 amino thiazoline 4 carboxilic acid, with other minor metabolites.
The body has several mechanisms to effectively detoxify cyanide. The majority of cyanide reacts with thiosulfate to produce thiocyanate in reactions catalyzed by sulfur tranferase enzymes such as rhodanese. The thiocyanate is then excreted in the urine over a period of days. Although thiocyanate is approximately seven times less toxic than cyanide, increased thiocyanate concentrations in the body resulting from chronic cyanide exposure can adversely affect the thyroid. Cyanide has a greater affinity for methemoglobin than for cytochrome oxidase, and will preferentially form cyanomethemoglobin. If these and other detoxification mechanisms are not overwhelmed by the concentration and duration of cyanide exposure, they can prevent an acute cyanide-poisoning incident from being fatal.
Some of the available antidotes to cyanide poisoning take advantage of these natural detoxifying mechanisms. Sodium thiosulfate, administered intravenously, provides sulfur to enhance the sulfur transferase-mediated transformation of cyanide to thiocyanate. Amyl nitrite, sodium nitrite and dimethyl aminophenol (DMAP) are used to increase the amount of methemoglobin in the blood, which then binds with cyanide to form non-toxic cyanomethemoglobin. Cobalt compounds are also used to form stable, non-toxic cyanide complexes, but as with nitrite and DMAP, cobalt itself is toxic.
Cyanide does not accumulate or biomagnify, so chronic exposure to sublethal concentrations of cyanide does not appear to result in acute toxicity. However, chronic cyanide poisoning has been observed in individuals whose diet includes significant amounts of cyanogenic plants such as cassava. Chronic cyanide exposure is linked to demyelination, lesions of the optic nerve, ataxia, hypertonia, Leber's optic atrophy, goiters and depressed thyroid function.
There is no evidence that chronic cyanide exposure has teratogenic, mutagenic or carcinogenic effects.
Cyanide in the Environment
Cyanide is produced naturally in the environment by various bacteria, algae, fungi and numerous species of plants including beans (chickpeas and lima), fruits (seeds and pits of apple, cherry, pear, apricot, peach and plum), almond and cashew nuts, vegetables of the cabbage family, grains (alfalfa and sorghum), roots (cassava, potato, radish and turnip), white clover and young bamboo shoots. Incomplete combustion during forest fires is believed to be a major environmental source of cyanide, and incomplete combustion of articles containing nylon produces cyanide through depolymerization.
Once released in the environment, the reactivity of cyanide provides numerous pathways for its degradation and attenuation:
Complexation: Cyanide forms ionic complexes of varying stability with many metals. Most cyanide complexes are much less toxic than cyanide, but weak acid dissociable complexes such as those of copper and zinc are relatively unstable and will release cyanide back to the environment. Iron cyanide complexes are of particular importance due to the abundance of iron typically available in soils and the extreme stability of this complex under most environmental conditions. However, iron cyanides are subject to photochemical decomposition and will release cyanide if exposed to ultraviolet light. Metal cyanide complexes are also subject to other reactions that reduce cyanide concentrations in the environment, as described below.
Precipitation: Iron cyanide complexes form insoluble precipitates with iron, copper, nickel, manganese, lead, zinc, cadmium, tin and silver. Iron cyanide forms precipitates with iron, copper, magnesium, cadmium and zinc over a pH range of 2-11.
Adsorption: Cyanide and cyanide-metal complexes are adsorbed on organic and inorganic constituents in soil, including oxides of aluminum, iron and manganese, certain types of clays, feldspars and organic carbon. Although the strength of cyanide retention on inorganic materials is unclear, cyanide is strongly bound to organic matter.
Cyanate: Oxidation of cyanide to less toxic cyanate normally requires a strong oxidizing agent such as ozone, hydrogen peroxide or hypochlorite. However, adsorption of cyanide on both organic and inorganic materials in the soil appears to promote its oxidation under natural conditions.
Thiocyanate: Cyanide reacts with some sulfur species to form less toxic thiocyanate. Potential sulfur sources include free sulfur and sulfide minerals such as chalcopyrite (CuFeS2), chalcocite (Cu2S) and pyrrhotite (FeS), as well as their oxidation products, such as polysulfides and thiosulfate.
Volatilization: At the pH typical of environmental systems, free cyanide will be predominately in the form of hydrogen cyanide, with gaseous hydrogen cyanide evolving slowly over time. The amount of cyanide lost through this pathway increases with decreasing pH, increased aeration of solution and with increasing temperature. Cyanide is also lost through volatilization from soil surfaces.
Biodegradation: Under aerobic conditions, microbial activity can degrade cyanide to ammonia, which then oxidizes to nitrate. This process has been shown effective with cyanide concentrations of up to 200 parts per million. Although biological degradation also occurs under anaerobic conditions, cyanide concentrations greater than 2 parts per million are toxic to these microorganisms.
Hydrolysis: Hydrogen cyanide can be hydrolyzed to formic acid or ammonium formate. Although this reaction is not rapid, it may be of significance in ground water where anaerobic conditions exist.
Effects on Wildlife: Although cyanide reacts readily in the environment and degrades or forms complexes and salts of varying stabilities, it is toxic to many living organisms at very low concentrations.
Aquatic Organisms: Fish and aquatic invertebrates are particularly sensitive to cyanide exposure. Concentrations of free cyanide in the aquatic environment ranging from 5.0 to 7.2 micrograms per liter reduce swimming performance and inhibit reproduction in many species of fish. Other adverse effects include delayed mortality, pathology, susceptibility to predation, disrupted respiration, osmoregulatory disturbances and altered growth patterns. Concentrations of 20 to 76 micrograms per liter free cyanide cause the death of many species, and concentrations in excess of 200 micrograms per liter are rapidly toxic to most species of fish. Invertebrates experience adverse nonlethal effects at 18 to 43 micrograms per liter free cyanide, and lethal effects at 30 to 100 micrograms per liter (although concentrations in the range of 3 to 7 micrograms per liter caused death in the amphipod Gammarus pulex).
Algae and macrophytes can tolerate much higher environmental concentrations of free cyanide than fish and invertebrates, and do not exhibit adverse effects at 160 micrograms per liter or more. Aquatic plants are unaffected by cyanide at concentrations that are lethal to most species of freshwater and marine fish and invertebrates. However, differing sensitivities to cyanide can result in changes to plant community structure, with cyanide exposures leaving a plant community dominated by less sensitive species.
The toxicity of cyanide to aquatic life is probably caused by hydrogen cyanide that has ionized, dissociated or photochemically decomposed from compounds containing cyanide. Toxic effects of the cyanide ion itself on aquatic organisms are not believed to be significant, nor are the effects of photolysis of ferro- and ferricyanides. It is therefore the hydrogen cyanide concentration of water that is of greatest significance in determining toxicity to aquatic life rather than the total cyanide concentration.
The sensitivity of aquatic organisms to cyanide is highly species specific, and is also affected by water pH, temperature and oxygen content, as well as the life stage and condition of the organism.
Birds: Reported oral LD50 for birds range from 0.8 milligrams per kilogram of body weight (American racing pigeon) to 11.1 milligrams per kilogram of body weight (domestic chickens). Symptoms including panting, eye blinking, salivation and lethargy appear within one-half to five minutes after ingestion in more sensitive species, and up to ten minutes after ingestion by more resistant species. Exposures to high doses resulted in deep, labored breathing followed by gasping and shallow intermittent breathing in all species. Mortality typically occurred in 15 to 30 minutes; however birds that survived for one hour frequently recovered, possibly due to the rapid metabolism of cyanide to thiocyanate and its subsequent excretion.
Ingestion of WAD cyanide solutions by birds my cause delayed mortality. It appears that birds may drink water containing WAD cyanide that is not immediately fatal, but which breaks down in the acidic conditions in the stomach and produces sufficiently high cyanide concentrations to be toxic.
Sublethal effects of cyanide exposure to birds, such as an increase in their susceptibility to predators, have not been fully investigated and reported.
Mammals: Cyanide toxicity to mammals is relatively common due to the large number of cyanogenic forage plants such as sorghum, sudan grasses and corn. Concentrations of cyanide in these plants are typically highest in the spring during blooming. Dry growing conditions enhance the accumulation of cyanogenic glycosides in certain plants as well as increase the use of these plants as forage.
Reported oral LD50 for mammals range from 2.1 milligrams per kilogram of body weight (coyote) to 6.0-10.0 milligrams per kilogram of body weight (laboratory white rats). Symptoms of acute poisoning usually occur within ten minutes of ingestion, including: initial excitability with muscle tremors; salivation; lacrimation; defecation; urination; labored breathing; followed by muscular incoordination, gasping and convulsions. In general, cyanide sensitivity for common livestock decreases from cattle to sheep to horses to pigs; deer and elk appear to be relatively resistant.
Although present in the environment and available in many plant species, cyanide toxicity is not widespread due to a number of significant factors:
Cyanide has low persistence in the environment and is not accumulated or stored in any mammal studied.
There is no reported biomagnification of cyanide in the food chain.
Although chronic cyanide intoxication exists, cyanide has a low chronic toxicity. Repeated sublethal doses of cyanide seldom result in cumulative adverse effects. Many species can tolerate cyanide in substantial yet sublethal intermittent doses for long periods of time.
Cyanide may be a major contributor to the morbidity and mortality observed in the approximately 5000-10,000 deaths from smoke inhalation occurring each year in the United States. Suicidal exposures are rarely reported to poison centers: intentional exposures accounted for 19 of the 187 cyanide poisoning cases reported to the American Association of Poison Control Centers in 2014.[17] However, a rapidly fatal suicide from cyanide salts in an adult patient might easily be mistaken for sudden death from myocardial infarction, pulmonary embolus, or ventricular dysrhythmia.
Suicide by cyanide poisoning occurs predominantly in males, as does industrial exposure. Leber optic atrophy has shown a very strong male predominance in European studies.
Deliberate ingestion of cyanide occurs mostly in adults. Smoke inhalation and chronic cyanide poisoning affect all ages.
Facts About Cyanide
What cyanide is
- Cyanide is a rapidly acting, potentially deadly chemical that can exist in various forms.
- Cyanide can be a colorless gas, such as hydrogen cyanide (HCN) or cyanogen chloride (CNCl), or a crystal form such as sodium cyanide (NaCN) or potassium cyanide (KCN).
- Cyanide sometimes is described as having a “bitter almond” smell, but it does not always give off an odor, and not everyone can detect this odor.
- Cyanide is also known by the military designations AC (for hydrogen cyanide) and CK (for cyanogen chloride).
Where cyanide is found and how it is used
- Cyanide is released from natural substances in some foods and in certain plants such as cassava, lima beans and almonds. Pits and seeds of common fruits, such as apricots, apples, and peaches, may have substantial amounts of chemicals which are metabolized to cyanide. The edible parts of these plants contain much lower amounts of these chemicals.
- Cyanide is contained in cigarette smoke and the combustion products of synthetic materials such as plastics. Combustion products are substances given off when things burn.
- In manufacturing, cyanide is used to make paper, textiles, and plastics. It is present in the chemicals used to develop photographs. Cyanide salts are used in metallurgy for electroplating, metal cleaning, and removing gold from its ore. Cyanide gas is used to exterminate pests and vermin in ships and buildings.
- If accidentally swallowed, chemicals found in acetonitrile-based products that are used to remove artificial nails can produce cyanide when metabolized by the body.
- Hydrogen cyanide, under the name Zyklon B, was used as a genocidal agent by the Germans in World War II.
- Reports have indicated that during the Iran-Iraq War in the 1980s, hydrogen cyanide gas may have been used along with other chemical agents against the inhabitants of the Kurdish city of Halabja in northern Iraq.
How you could be exposed to cyanide
- You could be exposed to cyanide by breathing air, drinking water, eating food, or touching soil that contains cyanide.
- Cyanide enters water, soil, or air as a result of both natural processes and industrial activities. When present in air, it is usually in the form of gaseous hydrogen cyanide.
- Smoking cigarettes is probably one of the major sources of cyanide exposure for people who do not work in cyanide-related industries.
How cyanide works
- The extent of poisoning caused by cyanide depends on the amount of cyanide a person is exposed to, the route of exposure, and the length of time that a person is exposed.
- Breathing cyanide gas causes the most harm, but swallowing cyanide can be toxic as well.
- Cyanide gas is most dangerous in enclosed places where the gas will be trapped.
- Cyanide gas evaporates and disperses quickly in open spaces, making it less harmful outdoors.
- Cyanide gas is less dense than air; so it will rise.
- Cyanide prevents the cells of the body from using oxygen. When this happens, the cells die.
- Cyanide is more harmful to the heart and brain than to other organs because the heart and brain use a lot of oxygen.
Immediate signs and symptoms of exposure to cyanide
- People exposed to a small amount of cyanide by breathing it, absorbing it through their skin, or eating foods that contain it may have some or all of the following signs and symptoms within minutes:
- Dizziness
- Headache
- Nausea and vomiting
- Rapid breathing
- Rapid heart rate
- Restlessness
- Weakness
- Exposure to a large amount of cyanide by any route may cause these other health effects as well:
- Convulsions
- Loss of consciousness
- Low blood pressure
- Lung injury
- Respiratory failure leading to death
- Slow heart rate
- Showing these signs and symptoms does not necessarily mean that a person has been exposed to cyanide.
Long-term health effects of exposure to cyanide
Survivors of serious cyanide poisoning may develop heart, brain and nerve damage.
How you can protect yourself, and what to do if you are exposed to cyanide
- Since breathing it is likely to be the primary route of exposure to cyanide, leave the area where the cyanide gas was released and get to fresh air. Quickly moving to an area where fresh air is available is highly effective in reducing exposure to cyanide gas.
- If the cyanide gas was released outdoors, move away from the area where it was released.
- If you cannot get out of the area where the cyanide gas was released, stay as low to the ground as possible.
- If the release of cyanide gas was indoors, get out of the building.
- If you are near a release of cyanide gas, emergency coordinators may tell you to either evacuate the area or “shelter in place” (stay put and take cover) inside a building to avoid being exposed to the chemical. For more information on evacuation during a chemical emergency, see Facts About Evacuation. For more information on sheltering in place during a chemical emergency, see Facts About Sheltering in Place.
- If you think you may have been exposed to cyanide, you should remove your clothing, rapidly wash your entire body with soap and water, and get medical care as quickly as possible.
- Removing your clothing:
- Quickly take off clothing that may have cyanide on it. Any clothing that has to be pulled over the head should be cut off the body instead of pulled over the head.
- If you are helping other people remove their clothing, try to avoid touching any contaminated areas, and remove the clothing as quickly as possible.
- Washing yourself:
- As quickly as possible, wash any cyanide from your skin with large amounts of soap and water. Washing with soap and water will help protect people from any chemicals on their bodies.
- If your eyes are burning or your vision is blurred, rinse your eyes with plain water for 10 to 15 minutes. If you wear contacts, remove them and put them with the contaminated clothing. Do not put the contacts back in your eyes (even if they are not disposable contacts). If you wear eyeglasses, wash them with soap and water. You can put your eyeglasses back on after you wash them. If you are wearing jewelry that you can wash with soap and water, you can wash it and put it back on. If it cannot be washed, it should be put with the contaminated clothing.
- Disposing of your clothes:
- After you have washed yourself, place your clothing inside a plastic bag. Avoid touching contaminated areas of the clothing. If you can't avoid touching contaminated areas, or you aren't sure where the contaminated areas are, wear rubber gloves or turn the bag inside out and use it to pick up the clothes, inverting the bag over the clothes when you have all the clothes picked up. An alternative method is to put the clothes in the bag using tongs, tool handles, sticks, or similar objects. Anything that touches the contaminated clothing should also be placed in the bag. If you wear contacts, put them in the plastic bag, too.
- Seal the bag, and then seal that bag inside another plastic bag. Disposing of your clothing in this way will help protect you and other people from any chemicals that might be on your clothes.
- When the local or state health department or emergency personnel arrive, tell them what you did with your clothes. The health department or emergency personnel will arrange for further disposal. Do not handle the plastic bags yourself.
- For more information about cleaning your body and disposing of your clothes after a chemical release, see Chemical Agents: Facts About Personal Cleaning and Disposal of Contaminated Clothing.
- Seek medical attention right away. Dial 911 and explain what has happened.
How cyanide poisoning is treated
Cyanide poisoning is treated with specific antidotes and supportive medical care in a hospital setting. Antidotes for cyanide poisoning are most useful if given as soon as possible after exposure. Clinicians should treat suspected cases accordingly and not wait for laboratory confirmation. The most important thing is for victims to seek medical treatment as soon as possible.
Prognosis
The prognosis in cyanide toxicity is good for patients who have only minor symptoms that do not necessitate the administration of antidotes. The prognosis is reasonably good for patients with moderate symptoms if rapid supportive intervention and effective antidotal therapy are provided. Suicidal poisonings tend to have severe outcomes because large doses are often involved.
The prognosis in patients with cyanogen poisoning is better in those with low-level exposures with minor symptoms that resolve after they are removed from exposure. The prognosis is fair for patients with seizures or recent-onset apnea if antidotes can be administered rapidly. The prognosis is generally poor in patients who suffer cardiac arrest secondary to cyanide toxicity, even if antidotes are administered promptly.
Mortality/morbidity
According to the American Association of Poison Control Centers Toxic Exposure Surveillance System, 7 of the 202 cyanide exposure cases in 2012 were fatal.[17]Cyanide induces fatality in seconds to minutes following inhalation or intravenous injection, in minutes following ingestion of soluble salts, or minutes (hydrogen cyanide) to several hours (cyanogens) after skin absorption.
Individuals who survive cyanide poisoning are at risk for central nervous system dysfunction, such as anoxic encephalopathy. Acute and delayed neurologic manifestations (Parkinson-like syndrome, other movement disorders, neuropsychiatric sequelae) have been reported.
Cyanides comprise a wide range of compounds of varying degrees of chemical complexity, all of which contain a CN moiety, to which humans are exposed in gas, liquid, and solid form from a broad range of natural and anthropogenic sources. While many chemical forms of cyanide are used in industrial application or are present in the environment, the cyanide anion CN– is the primary toxic agent, regardless of origin.
Hydrogen cyanide is a colourless or pale blue liquid or gas with a faint bitter almond-like odour. Hydrogen cyanide is used primarily in the production of substances such as adiponitrile, methyl methacrylate, chelating agents, cyanuric chloride, methionine and its hydroxylated analogues, and sodium and potassium cyanide. Hydrogen cyanide is also used as a fumigant in ships, railroad cars, large buildings, grain silos, and flour mills, as well as in the fumigation of peas and seeds in vacuum chambers.
Other cyanides, such as sodium and potassium cyanide, are solid or crystalline hygroscopic salts widely used in ore extracting processes for the recovery of gold and silver, electroplating, case-hardening of steel, base metal flotation, metal degreasing, dyeing, printing, and photography. They are also widely used in the synthesis of organic and inorganic chemicals (e.g., nitriles, carboxylic acids, amides, esters, and amines; heavy metal cyanides) and in the production of chelating agents.
Anthropogenic sources of cyanide release to the environment are diverse. Releases to air include chemical manufacturing and processing industries, such as metallurgical industries and metal plating, and extraction of gold and silver from low-grade ores. Other sources include volatilization from cyanide wastes disposed of in landfills and waste ponds, emissions from municipal solid waste incinerators, biomass burning, fossil fuel combustion, including vehicle emissions, fumigation operations, and the production of coke or other coal carbonization procedures.
Hydrogen cyanide is formed during the incomplete combustion of nitrogen-containing polymers, such as certain plastics, polyurethanes, and wool. Hydrogen cyanide is present in cigarette smoke.
Non-point sources of cyanide released to water can result from runoff from cyanide-containing anti-caking salts used on roads, migration from landfills, and agricultural and atmospheric fallout and washout. Point sources of releases to water include discharges from gold mining plants, wastewater treatment works, iron and steel production, and organic chemical industries.
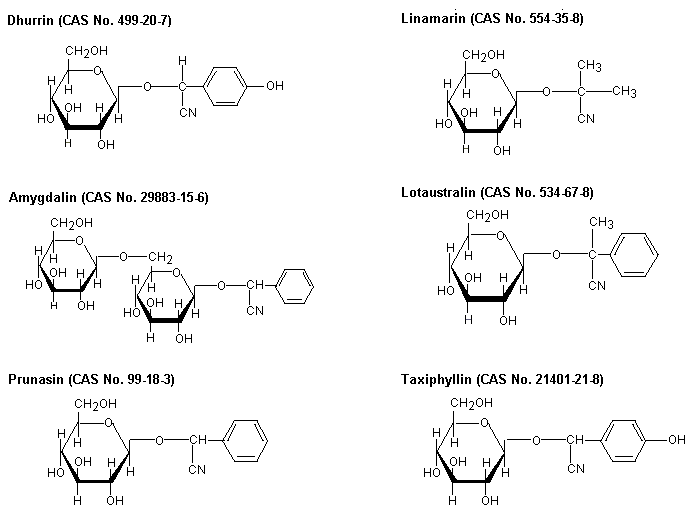
Principal natural sources of cyanides are over 2000 plant species, including fruits and vegetables, that contain cyanogenic glycosides, which can release cyanide on hydrolysis when ingested. Among them, cassava (tapioca, manioc) and sorghum are staple foods for hundreds of millions of people in many tropical countries. Known cyanogenic glycosides in plants include amygdalin, linamarin, prunasin, dhurrin, lotaustralin, and taxiphyllin. Hydrogen cyanide is released into the atmosphere from natural biogenic processes from higher plants, bacteria, and fungi.
In air, cyanide is present as gaseous hydrogen cyanide, with a small amount present in fine dust particles. Cyanides have the potential to be transported over long distances from their respective emission sources.
The majority of the population is exposed to very low levels of cyanide in the general environment. There are, however, specific subgroups with higher potential for exposure. These include individuals involved in large-scale processing of cassava and those consuming significant quantities of improperly prepared foods containing cyanogenic glycosides, such as cassava, speciality foods such as apricot pits, and bitter almonds. Other subgroups with greatest potential for exposure include those in the vicinity of accidental or intended releases from point sources, active and passive smokers, and fire-related smoke inhalation victims.
Workers may be exposed to cyanides during fumigation operations and the production and use of cyanides in many industrial processes — for example, electroplating, case-hardening of steel, and extraction of gold and silver from ores.
Cyanides are well absorbed via the gastrointestinal tract or skin and rapidly absorbed via the respiratory tract. Once absorbed, cyanide is rapidly and ubiquitously distributed throughout the body, although the highest levels are typically found in the liver, lungs, blood, and brain. There is no accumulation of cyanide in the blood or tissues following chronic or repeated exposure.
Approximately 80% of absorbed cyanide is metabolized to thiocyanate in the liver by the mitochondrial sulfur transferase enzyme rhodanese and other sulfur transferases. Thiocyanate is excreted in the urine. Minor pathways for cyanide detoxification involve reaction with cystine to produce aminothiazoline- and iminothiazolidinecarboxylic acids and combination with hydroxycobalamin (vitamin B12a) to form cyanocobalamin (vitamin B12); these end-products are also excreted in the urine.
The principal features of the toxicity profile for cyanide are its high acute toxicity by all routes of administration, with a very steep and rate-dependent dose–effect curve, and chronic toxicity, probably mediated through the main metabolite and detoxification product, thiocyanate. The toxic effects of cyanide ion in humans and animals are generally similar and are believed to result from inactivation of cytochrome oxidase and inhibition of cellular respiration and consequent histotoxic anoxia. The primary targets of cyanide toxicity in humans and animals are the cardiovascular, respiratory, and central nervous systems. The endocrine system is also a potential target for long-term toxicity, as a function of continued exposure to thiocyanate, which prevents the uptake of iodine in the thyroid and acts as a goitrogenic agent.
In humans, whereas slight effects occur at exposure levels of 20–40 mg/m3, 50–60 mg/m3 can be tolerated without immediate or late effects for 20 min to 1 h, 120–150 mg/m3 may lead to death after 0.5–1 h, 150 mg/m3 is likely to be fatal within 30 min, 200 mg/m3 is likely fatal after 10 min, and 300 mg/m3 is immediately fatal. The lowest reported oral lethal dose for humans is 0.54 mg/kg body weight, and the average absorbed dose at the time of death has been estimated at 1.4 mg/kg body weight (calculated as hydrogen cyanide). Sequelae after severe acute intoxications may include neuropsychiatric manifestations and Parkinson-type disease. Cyanide from tobacco smoke has been implicated as a contributing factor in tobacco–alcohol amblyopia. Long-term exposure to lower concentrations of cyanide in occupational settings can result in a variety of symptoms related to central nervous system effects.
Long-term consumption of cassava containing high levels of cyanogenic glycosides has been associated with tropical ataxic neuropathy, spastic paraparesis, and, in areas with low iodine intake, development of hypothyroidism, goitre, and cretinism. While exposure to cyanide has been crudely estimated to be 15–50 mg/day in endemic areas in some such cases, owing to the limitations of data on exposure and potential impact of confounders such as malnutrition, low protein content of the diet, vitamin deficiencies, and iodine status, the available data do not provide meaningful information on dose–response for cyanide.
Data on end-points other than acute toxicity are somewhat limited. This is attributable in large part to difficulties in conducting, for example, investigations of repeated-dose or chronic toxicity due to the high acute toxicity of the compound. Cyanides are weakly irritating to the skin and eye; data on sensitizing properties or carcinogenicity of hydrogen cyanide or its alkali salts have not been identified. Although somewhat limited, the weight of evidence of available data indicates that cyanide is not genotoxic and that it induces developmental effects only at doses or concentrations that are overtly toxic to the mothers.
Available data in human populations are considered inadequate as a basis for characterization of dose–response for chronic ingestion of cyanide. In a 13-week repeated-dose toxicity study in which cyanide was administered in drinking-water, there were no clinical signs associated with central nervous system effects or histopathological effects in the brain or thyroid of rats or mice exposed to doses up to 12.5 mg and 26 mg cyanide/kg body weight per day, respectively. At 12.5 mg cyanide/kg body weight per day, there were slight changes in the reproductive tract in male rats, which, although they apparently would not affect fertility in rats, are possibly significant to humans. The no-observed-adverse-effect level (NOAEL) for these effects was 4.5 mg/kg body weight per day. The examination of neurotoxicity in this study was limited to clinical observation and optical microscopy in autopsy. The few available studies specifically intended to investigate neurotoxicity, while reporting adverse effects at exposure levels of 1.2 mg cyanide/kg body weight per day in rats and 0.48 mg cyanide/kg body weight per day in goats, suffer from weaknesses that preclude their quantitative assessment.
In relation to characterization of concentration–response for repeated-dose toxicity for inhalation (relevant principally to the occupational environment), in three separate studies in rats, there were no adverse systemic effects in rats exposed to acetone cyanohydrin, which is rapidly hydrolysed to hydrogen cyanide at physiological pH, at concentrations up to 211 mg/m3 (corresponding to a concentration of 67 mg hydrogen cyanide/m3). The steepness of the dose–effect curve is illustrated by the observation of 30% mortality among rats exposed part of the day to 225 mg acetone cyanohydrin/m3 (71 mg hydrogen cyanide/m3).
Adverse effects of exposure to the low concentrations of cyanide that are generally present in the general environment (<1 µg/m3 in ambient air; <10 µg/litre in water) are unlikely. Acute cyanide intoxications may arise from eating apricot kernels, choke cherries, and other stone fruit kernels with high concentrations of cyanogenic glycosides. Inadequately prepared cassava, when constituting the major part of the diet, may be hazardous.
2. IDENTITY AND PHYSICAL/CHEMICAL PROPERTIES
Hydrogen cyanide (HCN) is a colourless or pale blue liquid or gas with a faint bitter almond-like odour. Common synonyms are hydrocyanic acid and prussic acid. Hydrogen cyanide is a very weak acid, with a pKa value of 9.22 at 25 °C. It is soluble in water and alcohol. Hydrogen cyanide is commercially available as a gas or as a technical-grade liquid in concentrations of 5, 10, and 96–99.5%. Phosphoric acid is added to liquid hydrogen cyanide as a stabilizer to prevent decomposition and explosion (ATSDR, 1997). Some important physical and chemical properties of hydrogen cyanide are summarized in Table 1.
The conversion factors2 for hydrogen cyanide in air (at 20 °C and 101.3 kPa) are as follows:
1 ppm = 1.12 mg/m3
1 mg/m3 = 0.890 ppm
Table 1: Physical and chemical properties of hydrogen cyanide (CAS No. 74-90-8 ).a
| Property | Value |
| Relative molecular mass | 27.03 |
| Boiling point (°C) | 25.70 |
| Solubility (30 °C) | Miscible with water; soluble in ethanol |
| Specific density: vapours (31 °C) | 0.937 |
| Odour threshold | 0.7 mg/m3 in air 0.17 mg/litre in water |
| Henry’s law constant (dimensionless) | 180–300b |
| Octanol/water partition coefficient (logKow) | 0.66 |
| Vapour pressure (kPa) | 35.2 at 0 °C 107.2 at 27.2 °C |
a From ACGIH (2001); DECOS (2002).
b Hine & Weimar (1965); Edwards et al. (1978); Gaffney et al. (1987).
Sodium cyanide (NaCN) is a white hygroscopic crystalline powder with a faint bitter almond-like odour. Common synonyms are cyanide of sodium and hydrocyanic acid, sodium. Commercially available sodium cyanide generally achieves a purity of 95–98%. The aqueous solution of sodium cyanide is strongly alkaline and rapidly decomposes. Sodium cyanide produces hydrogen cyanide on contact with acids or acid salts.
Potassium cyanide (KCN) is a white deliquescent solid with an odour of hydrogen cyanide. Common synonyms are hydrocyanic acid, potassium salt and cyanide of potassium. Potassium cyanide is commercially available at a 95% purity. An aqueous solution of potassium cyanide in water is strongly alkaline. Potassium cyanide also produces hydrogen cyanide on contact with acids or acid salts.
Calcium cyanide (Ca(CN)2), also commonly called cyanide of calcium, calcid, or calsyan, is a white crystalline solid. Its aqueous solution gradually liberates hydrogen cyanide. Cyanides such as sodium cyanide, potassium cyanide, and calcium cyanide form strong complexes with many metals (Table 2).
Cyanogen is a colourless toxic gas with an almond-like odour. Common synonyms are carbon nitrile, dicyanogen, ethane dinitrile, and oxalic acid dinitrile. Cyanogen is slowly hydrolysed in aqueous solution, yielding oxalic acid and ammonia. The conversion factors for cyanogen in air at 20 °C and 101.3 kPa are as follows:
1 ppm = 2.16 mg/m3
1 mg/m3 = 0.462 ppm
Table 2: Physical and chemical properties of selected cyanide compounds.a
| Species | CAS number | Molecular formula | Relative molecular mass | Common synonym(s) | Boiling point (°C) | Solubility |
| Sodium cyanide | NaCN | 49.02 | Cyanide of sodium | Soluble in water, slightly soluble in alcohol | ||
| Potassium cyanide | KCN | 65.11 | Cyanide of potassium | Soluble in water, slightly soluble in alcohol | ||
| Calcium cyanide | Ca(CN)2 | 92.12 | Calcid; calsyan | Soluble in water, slightly soluble in alcohol | ||
| Copper cyanide | 54-92-3 | CuCN | 89.56 | Cupricin | Insoluble in water | |
| Potassium silver cyanide | 501-61-6 | KAg(CN)2 | 198.01 | Potassium dicyanoargentate | Soluble in water, slightly soluble in ether | |
| Sodium ferrocyanide | Na4Fe(CN)6 | 303.91 | Sodium hexacyanoferrate (II) | Soluble in water | ||
| Potassium ferrocyanide | 13943-57-3 | K4Fe(CN)6 | 368.35 | Yellow prussiate of potash | Soluble in water | |
| Potassium ferricyanide | K3Fe(CN)6 | 329.95 | Red prussiate of potash | Slowly soluble in 2.5 parts of cold water; decomposes slowly in water | ||
| Cyanogen | NCCN | 52.04 | Carbon nitrile; dicyanogen | –20.7 | Soluble in water, alcohol, and ether | |
| Cyanogen chloride | CNCl | 61.47 | Chlorine cyanide | 13.8 | Soluble in water and alcohol | |
| Acetone cyanohydrin | (CH3)2C(OH)CN | 85.10 | ACH; methyllactonitrile | 82 | Soluble in water | |
| Sodium nitroprusside | Na2[Fe(CN)5NO] | 261.97 | Sodium nitroferrocyanide; sodium nitrosyl pentacyanoferrate (III) | Soluble in 2.3 parts of water, slightly soluble in alcohol |
a From Windholz (1983); ACGIH (2001); ECETOC (2004).
Cyanogen chloride is a colourless gas. Its common synonym is chlorine cyanide, and its common trade name is Caswell No. 267. Cyanogen chloride releases hydrogen cyanide by hydrolysis. Its conversion factors in air are:
1 ppm = 2.56 mg/m3
1 mg/m3 = 0.391 ppm
Common synonyms of acetone cyanohydrin are ACH, 2-cyano-2-propanol, 2-methyllactonitrile, and 2-hydroxy-2-methyl propanenitrile. It dissociates on standing to liberate hydrogen cyanide. Its boiling point is 120 °C (with decomposition to hydrogen cyanide and acetone). Its conversion factors in air are:
1 ppm = 3.54 mg/m3
1 mg/m3 = 0.283 ppm
The half-time of ACH in water was reported to be 9 min (Ellington et al., 1986); further studies reported that this hydrolysis to acetone and hydrogen cyanide was pH dependent, and half-times of 58, 27, and 8 min were observed at pH 4.8, 6.3, and 6.8 (ICI, 1993). In a more recent study, similar findings were reported (half-times of 54.7, 31.2, 5.4, and 4.0 min at pH 6.00, 6.40, 6.86, and 7.00, respectively) (Frank et al., 2002).
Some chemical properties of other cyanides are given in Table 2. Copper cyanide is a white to cream-coloured solid. Its common name is cuprous cyanide, and its synonym is cupricin. Potassium silver cyanide occurs as white crystals; its common synonym is potassium dicyanoargentate. It is sensitive to light. Sodium ferrocyanide decomposes at 435 °C, forming sodium cyanide.
Cyanogenic glycosides are produced naturally by many plants; when hydrolysed, they produce hydrogen cyanide. Chemical structures of some commonly occurring cyanogenic glycosides are depicted in Figure 1.
Further chemical and physical properties of hydrogen cyanide and some cyanides are summarized in the International Chemical Safety Cards included in this document.

Fig. 1: Cyanogenic glycosides in major edible plants (JECFA, 1993)
Amygdalin occurs in (among others) almonds, dhurrin in sorghum, linamarin in cassava, lotaustralin in cassava and lima beans, prunasin in stone fruits, and taxiphyllin in bamboo shoots.
3. ANALYTICAL METHODS
Cyanides in environmental media are usually collected in sodium or potassium hydroxide solution and measured by spectrophotometry (Agrawal et al., 1991), colorimetry, or ion-specific electrode or by headspace gas chromatography with a nitrogen-specific detector or electron capture detector (Maseda et al., 1989; Seto et al, 1993). Cyanide in aqueous matrices is usually measured by colorimetric, titrimetric (US EPA, 1983), or electrochemical methods after pretreatment to produce hydrogen cyanide and absorption in sodium hydroxide solution. Total cyanide (irrespective of origin) includes all of the available cyanide in a sample; in drinking-water, it is measured by semi-automated colorimetry (EPA Method 335.4) as well as by selective electrode, ultraviolet/distillation/spectrophotometry, and ion chromatography (EPA Method 300.0) (US EPA, 1993a). Free cyanide can also be determined by one method (SM-4500-CN-F) approved for drinking-water compliance monitoring analysis that does not require distillation, the specific ion electrode method (US EPA, 2003a). Weak acid dissociable cyanide analysis (used principally by the precious metals mining industry) includes those cyanide species liberated at moderate pH 4.5, such as aqueous hydrogen cyanide and cyanide anion, the majority of copper, cadmium, nickel, zinc, silver, and tin complexes, and others with similar low dissociation constants. Weak acid dissociable cyanide can be determined in wastewaters by a ligand exchange/flow injection/amperometric technique (EPA Method 1677) (Milosavlievic et al., 1995; US EPA, 1997).
A chromatographic technique with fluorescence detection is used to detect trace amounts of cyanide in blood cells (Chinaka et al., 1998). Cyanide in biological tissue and fluids can be measured spectrophotometrically after reaction with methaemoglobin.
Since many cyanides are unstable and emit volatile hydrogen cyanide gas, sampling, storage, and analysis must be done with caution, preferably immediately upon collection.
The three commonly used techniques (colorimetric, titrimetric, and electrochemical) may all suffer from interference problems, unless proper precautions are taken (ATSDR, 1989).
Metals suppress the transformation of cyanide to formic acid, thus lowering the measured hydrogen cyanide concentration (Dolzine et al., 1982). Carbonyl compounds also decrease the hydrogen cyanide recovery (Honig et al., 1983), as in the case with soybean samples, in which carbonyl compounds occur naturally.
Sodium thiosulfate can interfere with potentiometric (Sylvester et al., 1982) or colorimetric analysis (Ganjeloo et al., 1980). Care should be taken, since it is often used as an antidote to treat chemical poisoning.
Continuous monitoring of cyanide is also available using equipment based on diffusion and amperometric detection of hydrogen cyanide (NIOSH, 1976).
Detection limits for the different methods for hydrogen cyanide range from 0.8 to 400 mg/m3 for air samples, from 0.04 to 200 µg/litre for aqueous samples, and from 0.8 to 300 µg/litre for biological samples. NIOSH Method 7904 for workplace air has a limit of detection of 2.5 µg cyanide (NIOSH, 1994).
4. SOURCES OF HUMAN AND ENVIRONMENTAL EXPOSURE
4.1 Natural occurrence
Hydrogen cyanide is ubiquitous in nature. It is found in the stratosphere and non-urban troposphere (US EPA, 1990). It is released into the atmosphere from biomass burning, volcanoes, and natural biogenic processes from higher plants, bacteria, algae, and fungi (Fiksel et al., 1981; Cicerone & Zellner, 1983; Way, 1984; ATSDR, 1997; Li et al., 2000). An estimate of the amount of cyanide released to the environment from natural biogenic processes is not available (ATSDR, 1997).
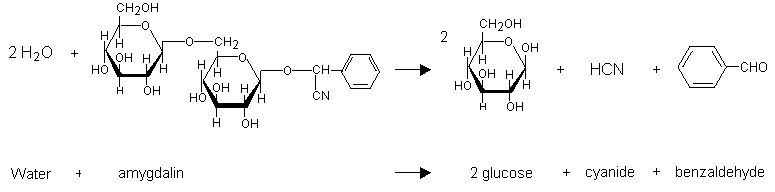
Cyanide occurs naturally as cyanogenic glycosides in at least 2000 plants (Figure 1). Amygdalin (d-mandelonitrile-beta-d-glucoside-6-beta-d-glucoside) has been found in about 1000 species of plants, including cassava (tapioca, manioc), sweet potato, corn, cabbage, linseed, millet, and bamboo, in pits of stone fruits, such as cherries, peaches, and apricots, and in apple seeds (JECFA, 1993; Sharma, 1993; Padmaja, 1995). It is also present in bitter almonds and American white lima beans (Ermans et al., 1972). After ingestion, linamarin can be hydrolysed by either cassava linamarase or an endogenous beta-glucosidase to yield d-glucose and ACH (Frakes et al., 1986a).
4.2 Anthropogenic sources
4.2.1 Production
Hydrogen cyanide is principally produced by two synthetic catalytic processes involving the reaction of ammonia and natural gas (or methane) with or without air. It is also obtained as a by-product in the production of acrylonitrile by the ammoxidation of propylene, which accounts for approximately 30% of the worldwide production of hydrogen cyanide.
Sodium and potassium cyanides are principally prepared by the direct reaction of hydrogen cyanide with the respective alkali in closed systems (European Chemicals Bureau, 2000a,b). Sodium cyanide is also prepared to a lesser extent by melting sodium chloride with calcium cyanamide or by heating sodium amide salt with carbon.
Calcium cyanide is produced by the reaction of coke, coal, and limestone.
Cyanogen chloride is a reaction product of organic precursors with hypochlorous acid in the presence of ammonia and may be formed as a by-product of the chloramination of water (WHO, 1996; IPCS, 2000a).
ACH was first produced in the 1930s as an intermediate in the production of methyl methacrylate from hydrogen cyanide. It is currently produced from the liquid-phase reaction of hydrogen cyanide and acetone in the presence of an alkali catalyst at atmospheric pressure (ECETOC, 2004).
Hydrogen cyanide capacity is generally treated as the sum of purposeful direct synthesis and that derived as a by-product of acrylonitrile production. Annual US hydrogen cyanide capacity by 11 companies in 1991 was 666 000 tonnes. US production of hydrogen cyanide from 1983 to 1989 rose from 300 000 to 445 000 tonnes (Pesce, 1993). Output of hydrogen cyanide in the USA was 545 000 tonnes in 1992 (Cohrssen, 2001). Worldwide annual production and capacity of hydrogen cyanide in 1992 were estimated to be 950 000 and 1 320 000 tonnes, respectively (Pesce, 1993; Cohrssen, 2001). It has been estimated that the present total annual production of hydrogen cyanide worldwide is 1.4 million tonnes (Mudder & Botz, 2000).
4.2.2 Use
In 1983, the major end uses of hydrogen cyanide in the USA were in the production of adiponitrile (200 000 tonnes), ACH (128 000 tonnes), cyanuric chloride (28 500 tonnes), sodium cyanide (69 000 tonnes), chelating agents (15 800 tonnes), and nitrilotriacetic acid (10 100 tonnes) and for miscellaneous uses (20 000 tonnes) (US EPA, 1990). Hydrogen cyanide is also used in the production of methyl methacrylate, methionine and its hydroxylated analogues, and potassium cyanide (ATSDR, 1997; ECETOC, 2004).
Sodium cyanide is extensively employed in a large number of industrial processes, including electroplating and case-hardening of metals; the extraction (cyanidation) of gold and silver from ores; base metal flotation; coal gasification; and the fumigation of ships, railroad cars, buildings, grain silos, flour mills, seeds in vacuum chambers, and soil. Large quantities of sodium cyanide are used to introduce cyano groups into organic compounds, in particular through a reaction with organic halogen compounds to yield nitriles. The nitriles can then be converted to a variety of carboxylic acids, amides, esters, and amines. Potassium cyanide is used for electrolytic refining of platinum, for metal colouring, and as an electrolyte for the separation of gold, silver, and copper from platinum (Eisler et al., 1999; Patnaik, 1999; ACGIH, 2001; ECETOC, 2004). Cyanide salts are used as chelating agents, and the complex cyanides of copper, zinc, and cadmium are used in electroplating processes, principally the plating of iron, steel, and zinc (ECETOC, 2004).
Calcium cyanide is used chiefly as a fumigant, because it readily releases hydrogen cyanide when exposed to air; as a fertilizer, defoliant, herbicide, and rodenticide; as a stabilizer for cement; and in stainless steel manufacture (ACGIH, 2001).
Cyanogen is used as a fumigant, as a fuel gas for welding and cutting heat-resistant metals, and as a rocket and missile propellant (ATSDR, 1997).
Cyanogen chloride is used as a fumigant gas and as a reagent in chemical synthesis.
Cuprous cyanide is used in plating baths for silver, brass, and copper–tin alloy plating (ATSDR, 1997), as an antifouling agent in marine paint, and as an insecticide and fungicide (Windholz, 1983).
Potassium silver cyanide is used in silver plating and as a bactericide.
Potassium ferricyanide is used chiefly for blueprints, in photography, for staining wood, in calico printing, and in electroplating.
Sodium ferrocyanide is used in ore flotation, as an anti-caking agent in rock salt, and in photography for bleaching, toning, and fixing.
Sodium nitroprusside has been used as an antihypertensive agent and in congestive heart failure and is used for deliberate induction of hypotension during certain neurosurgical procedures.
ACH is used in preparative transcyanohydrination reactions.
4.2.3 Release to the environment
More than 30 large-scale accidental releases of cyanide to water systems have been reported since 1975; these include transportation accidents, pipe failures, and tailings dam-related releases (Korte et al., 2000; Mudder & Botz, 2000).
Non-point sources of cyanide released to water can result from runoff from cyanide-containing anti-caking salts (i.e., sodium ferrocyanide) used on roads, migration from landfills, and agricultural and atmospheric fallout and washout (ATSDR, 1997).
The extraction of gold from low-grade ores by cyanidation processes was estimated to result in a worldwide emission of 20 000 tonnes of hydrogen cyanide into the atmosphere (Korte & Coulston, 1998). Another estimate suggested that currently 45 300 tonnes of cyanide are used in the USA in the cyanidation process. The wastes from these processes result in large cyanide-containing ponds near the mining operations (Clark & Hothem, 1991; Henny et al., 1994; Ma & Pritsos, 1997; Eisler et al., 1999).
The major point sources of cyanide release to water are discharges from gold mining plants, publicly owned wastewater treatment plants, iron and steel production, and the organic chemical industries. An estimated 3 billion litres (i.e., 3 × 109 litres) of wastes containing cyanides were generated in the USA in 1983, principally from spent cyanide plating bath solutions from electroplating operations (except for precious metals) and from spent stripping and cleaning bath solutions from electroplating operations (Grosse, 1986).
During cassava starch production, large amounts of cyanoglycosides are released and hydrolysed by plant-borne enzymes, leading to cyanide concentrations in wastewater as high as 200 mg/litre (Siller & Winter, 1998).
The major sources of cyanide released to air, in addition to exhaust from vehicle emissions, are diverse, including chemical manufacturing (hydrogen cyanide, methyl methacrylate, acrylonitrile); processing industries, such as metallurgical industries and metal plating (i.e., electroplating metals and finishing [metal polishes]); extraction of gold and silver from low-grade ores; volatilization from cyanide wastes disposed of in landfills and waste ponds; the production of coke or other coal carbonization procedures; emissions from municipal solid waste incinerators; and direct release of cyanides to the atmosphere resulting from fumigation operations, combustion of polyurethanes, acrylonitrile, and polyamide plastics, and combustion of wool, silk, and fibres (Carotti & Kaiser, 1972; Fiksel et al., 1981; ATSDR, 1997; Eisler et al., 1999).
An estimated total of 1 million tonnes of hydrogen cyanide, amounting to 73.1% of the total environmental releases in the USA, was discharged to the air from manufacturing and processing facilities (ATSDR, 1997).
The estimated amounts of hydrogen cyanide released to air in 1976 from the most common non-industrial sources were as follows: agricultural pest control, 62 tonnes; incineration, 8.2–82 tonnes; and tobacco smoke, 5.9–340 tonnes (Fiksel et al., 1981; ATSDR, 1997).
In 2001, from various locations in the USA, about 1300 tonnes of hydrogen cyanide were released on- and off-site; 540 tonnes were emitted to the atmosphere, 0.1 tonne was released to surface waters, 770 tonnes were injected into Class I wells,3 and 0.42 tonne was released to land (US EPA, 2003c). In 2001, from various locations in the USA, approximately 3400 tonnes of cyanides (not otherwise specified) were released on- and off-site; 220 tonnes were emitted to the atmosphere, 47 tonnes were released to surface waters, 1800 tonnes were injected into Class I wells, and 1300 tonnes were released to land (US EPA, 2003c).
Hydrogen cyanide has been found following the combustion of a number of synthetic polymers. The maximum yield of hydrogen cyanide per gram of polyurethane foam ranged from 0.37 to 0.93 mg under non-flaming conditions and from 0.5 to 1.02 mg under flaming combustion (Sklarew & Hayes, 1984). Hydrogen cyanide concentrations in the off-gas from the shale oil retorting process ranged from 7 to 44 mg/m3 (Sklarew & Hayes, 1984).
One cigarette without a filter liberates 500 µg hydrogen cyanide, while filter cigarettes liberate only 100 µg in mainstream smoke. Hydrogen cyanide concentrations in mainstream and sidestream smoke ranging from 280 to 550 µg/cigarette and from 53 to 111 µg/cigarette, respectively, have been reported; sidestream:mainstream ratios of hydrogen cyanide concentrations ranged from 0.06 to 0.50 (ATSDR, 1997). The level of hydrogen cyanide found in Canadian cigarette smoke under International Organization for Standardization standard smoking conditions were as follows: mainstream smoke, 32–156 µg/cigarette; and sidestream smoke, 77–136 µg/cigarette (Health Canada, 2002).
The average rate of emission of hydrogen cyanide by automobile exhaust was reported to be 7–9 mg/km for cars not equipped with catalytic converters and on the order of 0.6 mg/km for cars with catalytic converters operating under optimum conditions in the mid- to late 1970s (ATSDR, 1997).
Cyanogen chloride is formed as a reaction product of organic precursors with hypochlorous acid in the presence of ammonia and may be formed as a by-product of the chloramination of water (e.g., via the reaction of humic substances with chlorine and chloramine used for water disinfection) (Ohya & Kanno 1987; WHO, 1996; IPCS, 2000a). In the USA, 35% of the surface water plants and 23% of the groundwater plants using chloramine as a primary or secondary disinfectant report cyanogen chloride formation (US EPA, 2002).
Cyanogen is generated in the combustion of nitrogen–carbon compounds and appears in automobile exhaust gases and gases from blast furnaces (CHEMINFO, 1998).
Cyanide is present in the air mostly as a gas, and cyanides have the potential to be transported over long distances from their respective emission sources.
5. ENVIRONMENTAL LEVELS AND HUMAN EXPOSURE
5.1 Environmental levels
5.1.1 Air
Cyanide is found in ambient air as hydrogen cyanide and to a smaller extent in particulate matter. The concentration of hydrogen cyanide measured since 1981 in the northern hemisphere’s non-urban troposphere ranged from 180 to 190 ng/m3 (Cicerone & Zellner, 1983; Jaramillo et al., 1989).
Ambient air monitoring data for cyanides in Bulgaria in areas near petrochemical plants showed concentrations ranging from 0.2 to 0.8 µg/m3 (annual average value) (Kaloyanova et al., 1985).
Cyanide has been detected at levels of 20–46 mg/m3 in the air near large-scale cassava processing facilities in Nigeria (Okafor & Maduagwu, 2000).
5.1.2 Water
Cyanides, reported as cyanide, hydrogen cyanide, sodium cyanide, potassium cyanide, calcium cyanide, or copper(I) cyanide, have been detected in surface water samples at 70 of the 154 hazardous waste sites where they were studied in the USA; they have also been detected in groundwater samples at 191 of the 419 waste sites studied and in leachate samples of 16 of the 52 sites studied. The median concentrations in the positive samples were 160 µg/litre for groundwater, 70 µg/litre for surface water, and 479 µg/litre for the leachates (HazDat, 2003).
Data from the US National Urban Runoff Program in 1982 revealed that 16% of urban runoff samples collected from four cities across the USA contained cyanides at levels of 2–33 µg/litre (ATSDR, 1997).
According to the US Environmental Protection Agency’s (EPA) STORET database, the mean cyanide concentration in most surface waters in the USA is less than 3.5 µg/litre. Data from the late 1970s to early 1980s indicated that the levels are higher only in limited areas and may exceed 200 µg/litre (ATSDR, 1997).
In 1978, a US EPA survey of drinking-water supplies showed that about 7% of the supplies had cyanide concentrations greater than 10 µg/litre (US EPA, 1993a). Cyanogen chloride is one of the 18 compounds that occur most frequently (8 of 10 city surveys) in potable water within the framework of the US National Organic Reconnaissance Survey (Bedding et al., 1982). In a survey in 1987 of over 35 drinking-water supplies, the quarterly median cyanogen chloride concentrations in drinking-water ranged from 0.45 to 0.80 µg/litre (from 0.19 to 0.34 µg cyanide/litre) (Krasner et al., 1989; ATSDR, 1997). More current data regarding the cyanide and cyanogen chloride levels in drinking-water are lacking.
Levels of 1.58–7.89 mg cyanide/litre have been found in natural water sources near large-scale cassava processing facilities in Nigeria (Okafor et al., 2001).
5.1.3 Soil
Cyanide has been identified in the soil of hazardous waste sites in the USA; the median concentrations for the positive sites were 0.8 mg/kg in the subsurface soil (found at 77 sites of the 124 studied) and 0.4 mg/kg in the topsoil (51 positive sites out of 91 sites) (HazDat, 2003).
Cyanide-containing wastes are commonly found in soils at former manufactured gas plant sites in the USA. Most concentrations of cyanide compounds at the manufactured gas plant sites are below 2000 mg/kg. The most prevalent types of cyanide compounds are iron-complexed forms, e.g., ferric ferrocyanide (Prussian blue), rather than the highly toxic free cyanide forms. Iron-complexed cyanides, dominated by the ferrocyanide ion, comprise over 97% of total cyanides in either weathered or unweathered soils (Shifrin et al., 1996).
5.1.4 Food
Many edible plants contain cyanogenic glycosides, whose concentrations can vary widely as a result of genetic and environmental factors, location, season, and soil types (Ermans et al., 1980; JECFA, 1993). Some of the foodstuffs and their cyanide contents are shown in Table 3. Cassava tubers vary widely in their cyanogenic glycoside content, although most varieties contain 15–400 mg cyanide/kg fresh weight. Occasionally varieties of cassava tubers contain 1300–2000 mg cyanide/kg fresh weight, and cassava leaves contain 1000–2000 mg cyanogenic glucosides/kg on a dry matter basis (Padmaja, 1995). Fermentation of cassava pulp for 96 h during gari production reduced the hydrogen cyanide content by 50%; soaking of sliced cassava for 24 h, 40%; and sun-drying, some 15% (Kendirim et al., 1995). It should be noted that the ranges of cyanide concentrations shown in Table 3 are very broad in several cases (i.e., cereals and their products, soy protein products, and apricot pits), which may be due to their different sources and differences in analytical procedures; as well, the values may reflect the older literature.
Hydrogen cyanide can be produced by hydrolytic reaction catalysed by one or more enzymes from the plants containing cyanogenic glycosides. In kernels, for example, this reaction is catalysed by the enzyme emulsin (Lasch & El Shawa, 1981) when the seeds are crushed and moistened. Amygdalin (which is also present in cassava, bitter almonds, and peach stones) is converted to glucose, benzaldehyde, and hydrogen cyanide (Figure 2) (IPCS, 1992). Hydrogen cyanide release can occur during maceration, which activates intracellular beta-glucosidases. This reaction can also result from chewing, which causes the enzyme and the cyanogenic glycosides stored in different compartments to combine (Ermans et al., 1980; Nahrstedt, 1993). The reaction occurs rapidly in an alkaline environment, and the hydrolysis is complete in 10 min. Hydrolysis is possible in an acid solution and takes place slowly.

Fig. 2: Hydrolysis of amygdalin
Liberation of hydrogen cyanide from cyanogenic glycosides occurs usually after ingestion and hydrolysis by the glycosidases of the intestinal microflora and, to a lesser degree, by glucosidases of the liver and other tissues (Padmaja, 1995). However, hydrolysis may also occur during the preparation of the food, which may account for the short interval between ingestion and the appearance of signs of poisoning in some accidents (Lasch & El Shawa, 1981).
Table 3: Cyanide concentrations in food products.a
| Type of product | Cyanide concentration (in mg/kg or mg/litre) |
| Cereal grains and their products | 0.001–0.45 |
| Soy protein products | 0.07–0.3 |
| Soybean hulls | 1.24 |
| Apricot pits, wet weight | 89–2170 |
| Home-made cherry juice from pitted fruits | 5.1 |
| Home-made cherry juice containing 100% crushed pits | 23 |
| Commercial fruit juices | |
| Cherry | 4.6 |
| Apricot | 2.2 |
| Prune | 1.9 |
| Tropical foodstuffs | |
| Cassava (bitter) / dried root cortex | 2360 |
| Cassava (bitter) / leaves | 300 |
| Cassava (bitter) / whole tubers | 380 |
| Cassava (sweet) / leaves | 451 |
| Cassava (sweet) / whole tubers | 445 |
| Gari flour (Nigeria) | 10.6–22.1 |
| Sorghum / whole immature plant | 2400 |
| Bamboo / immature shoot tip | 7700 |
| Lima beans from Java (coloured) | 3000 |
| Lima beans fom Puerto Rico (black) | 2900 |
| Lima beans from Burma (white) | 2000 |
a From Nartey (1980); Honig et al. (1983); JECFA (1993); ATSDR (1997).
5.1.5 Other
Laetrile (another name for amygdalin derived from apricot kernels), which was formerly used as an anticancer agent, releases cyanide upon metabolism. Bitter almonds and apricot pits containing cyanogenic glycosides are still sold in health food stores and over the Internet (Suchard et al., 1998). Other drugs, such as sodium nitroprusside, which is used as an antihypertensive and in congestive heart failure (Guiha et al., 1974; Tinker, 1976; Aitken et al., 1977; Schultz, 1984; Rindone & Sloane 1992), also liberate hydrogen cyanide in the body. In sodium nitroprusside, the CN– moiety represents 44% by weight of the molecule. Some aliphatic nitriles that are widely used in the chemical industry — i.e., acetonitrile (IPCS, 1993), acrylonitrile (IARC, 1999), succinonitrile, and adiponitrile — also release cyanide upon metabolism (Willhite & Smith, 1981).
5.2 Human exposure
5.2.1 General population
The general population may be exposed to cyanide from ambient air, drinking-water, and food.
Based on an atmospheric hydrogen c
At a joint meeting of the World Federation of Associations ofClinical Toxicology and Poison Control Centres, the InternationalProgramme on Chemical Safety (IPCS), and the Commission of theHealth Organization in October 1985, the evaluation of antidotesEuropean Communities (CEC), held at the headquarters of the Worldarea for international collaboration. During 1986, the IPCS and CECused in the treatment of poisonings was identified as a prioritytherapeutic substance used to counteract the toxic action(s) of aundertook the preparatory phase of a joint project on this subject. For the purpose of the project an antidote was defined as ato treat their effects on body functions, were listed andspecified xenobiotic. Antidotes, as well as other agents used to prevent the absorption of poisons, to enhance their elimination andwere classified as: (1) those generally accepted as useful; (2)preliminarily classified according to the urgency of treatment and efficacy in practice. With respect to efficacy in practice, they those widely used and considered promising but not yet universallyused for specific purposes were considered to correspond to the WHOaccepted as useful and requiring further research concerning their efficacy and/or their indications for use; and (3) those of questionable usefulness. Additionally, certain antidotes or agents criteria for essential drugs (see Criteria for the Selection ofThese were included in volume 1 of this series.Essential Drugs, WHO Technical Report Series 722, Geneva, 1985). A methodology for the principles of evaluating antidotes and agents used in the treatment of poisonings and a proforma for preparing monographs on antidotes for specific toxins were drafted.necessary. Accordingly, several were selected for initial reviewMonographs are being prepared, using the proforma, for those antidotes and agents provisionally classified in category 1 as regards efficacy in practice. For those classified in categories 2 and 3, where there are insufficient data or controversy regarding efficacy in practice, it was agreed that further study wasToxicologists (EAPCCT; formerly known as the European Association ofand evaluation, among which were antidotes used in the treatment of poisoning by cyanide. The review and evaluation of antidotes used in the treatment of poisoning by cyanide was initiated at a joint meeting of the European Association of Poison Control Centres and Clinical Poison Control Centres), the IPCS, and the CEC, organized by thePersson, hydroxocobalamin by Professor C. Bismuth, dicobalt edetateNational Poison Information Centre of the Netherlands National Institute of Public Health and Environmental Hygiene and held at the University Hospital AZU, Utrecht, The Netherlands, 13-15 May 1987. In preparation for this meeting, documents were drafted, using the proforma, on oxygen by Dr U. Taitelman, sodium thiosulfate by Dr H. by Dr T.C. Marrs, sodium nitrite by Dr A. Hall, andNetherlands) on the clinical aspects of cyanide antidotes. The4-dimethylaminophenol by Professor M. von Clarmann. Also in preparation for the meeting, documents were drafted by Professor M. Geldmacher von Mallinckrodt on the analytical assessment of cyanide poisoning, by Dr A. van Dijk on the pharmaceutical aspects of cyanide antidotes, and by Professor A.N.P. van Heijst (formerly Director, Dutch National Poison Control Centre, Utrecht, thethe comments on the documents and of the additional materialdocuments presented by each author were discussed at the meeting and participants gave their own experience and views. Experience of industrial aspects of cyanide poisoning was presented by Dr A.C.G.M. Parren. The main meeting was followed by that of an IPCS/CEC working group, consisting of the authors of documents, the meeting rapporteur and a number of observers, at which a review was made ofmetabolic and haemoglobin abnormalities). The group concentrated onpresented at the main meeting. Based on the available material, an evaluation was made of the different approaches to treatment of cyanide poisoning depending on the type of cyanide exposure (hydrogen cyanide, either alone or with carbon monoxide, cyanide salts or cyanogenic glycosides), the state of intoxication and number of patients, the location of the patient with respect to treatment facilities, and special situations (e.g., inheritedantidotes for methaemoglobin-forming agents. Concerning theacute poisoning by cyanide, considering that there were insufficient data for evaluating approaches to treatment of chronic cyanide toxicity. Nevertheless, it was considered that a review of chronic poisoning by cyanide, particularly in relation to cyanide ingestion from food, was needed. It was agreed that traditional means of treatment of cyanide poisoning would have to be revised, and that any evaluation of approaches to treatment must also includeconsisting of Professor A.N.P. van Heijst (chairman of the meeting),analytical aspects, it was noted that there was particular difficulty in measuring the concentration of cyanide in blood if an antidote had already been administered, a problem that is being studied by a group of experts established under the auspices of the German Research Association Commission on Clinical Analytical Toxicology. A number of new cyanide antidotes in various stages of research and development were discussed. An editorial groupwas invited to the meeting but was unable to attend, prepared aDr T.J. Meredith (rapporteur), Dr J.A. Haines (IPCS, chairman of the working group) and Dr J.-C. Berger (CEC) was established in order to prepare a consolidated monograph on cyanide antidotes. Draft documents were revised by their authors. Those on methylene blue and toluidine blue were prepared by Dr Christina Alonzo (CIAT, Montevideo, Uruguay) and Dr T.C. Marrs, respectively. Subsequently Dr J.A. Vick (Food and Drug Administration, USA), who draft document on experience with the use of amyl nitrite inthe preparation and finalization of this monograph are gratefullytreating cyanide poisoning in animals. Professor C. Bismuth and Dr A. Hall drafted material on new antidotes under development for clinical trials, and Dr A.C.G.M. Parren drafted material on protective measures. The editorial group met twice in Utrecht on 22-23 October 1987 and 20-22 July 1988. Material was checked and rearranged, additional material was prepared for a number of the chapters and the overview chapter was drafted. The efforts of all who helped in acknowledged. ABBREVIATIONS ATA atmosphere absolute BE base excessOHB12 hydroxocobalaminCNS central nervous system CT computer tomography 4-DMAP 4-dimethylaminophenol EDTA ethylenediaminetetraacetic acid G6PD glucose-6-phosphate dehydrogenase Hb haemoglobin HMPS hexose monophosphate shunt INN international non-proprietary name LDLo lowest published lethal dose MLD minimal lethal dose NADH reduced nicotinamide adenine dinucleotide NADPH reduced nicotinamide adenine dinucleotide phosphateLD50 Lethal Dose 50USP United States PharmacopoeiaB12 Vitamin B12HbO2 OxyhaemoglobinAV atrioventricularSNP sodium nitroprusside VS volumetric solutionThe recognition of cyanide as a poison in bitter almonds,cherry laurel leaves, and cassava goes back to antiquity. Anrefers to the "penalty of the peach," and Dioscorides in the firstinscription on an Egyptian papyrus in the Louvre Museum, Paris,The first description of cyanide poisoning was by Wepfer incentury A.D. was aware of the poisonous properties of bitter almonds (Sykes, 1981).Ireland caused by drinking cherry laurel water, used as a flavouring1679 and dealt with the effects of the administration of extract of bitter almonds (Sykes, 1981). Two fatal cases of poisoning inpoison; given orally, into the rectum, or by injection, it rapidlyagent in cooking and to dilute brandy, led to the experiments of Madden (1731). He showed that cherry laurel water contains a killed dogs. It was not until 1786 that isolation of pure hydrogenSchrader (1802). The introduction of cyanide as a medicament tocyanide (HCN) from the dye Prussian blue was achieved by Scheele (1786). The mechanism of toxicity of cyanide was explored by Fontana (1795). Cyanide was obtained from bitter almonds by treat coughs and lung diseases was suggested by Magendie (1817).Hoppe-Seyler (1876) reported that cyanide inhibits tissue oxidationIndeed, it was not until 1948 that cherry laurel water was removed from the British Pharmacopoeia! Attempts to antagonize the toxic effects of cyanide were reported by Blake (1839 and 1840). reactions. Antagonism between amyl nitrite and prussic acid was mentionedChen et al. (1933, 1934). They suggested using a combination ofby Pedigo (1888), and, as early as 1894, cobalt compounds were advocated by Antal (1894) as cyanide antagonists. Sodium nitrite was used as an antidote in experimental cyanide poisoning by Mladoveanu & Gheorghiu (1929). A biochemical mechanism for cyanide antagonism was described bytoxicological reasoning. This combination of antidotes has stoodamyl nitrite, sodium nitrite and sodium thiosulfate, the latter compound serving as a sulfur donor for rhodanese (thiosulfate sulfur transferase). Rhodanese accelerates cyanide detoxification by forming the metabolite thiocyanate. This represented the development of one of the first antidotes based on scientificB12a) combined with cyanide to form cyanocobalamin (vitaminthe test of time, and still represents one of the most efficacious antidotal combinations for the treatment of cyanide intoxication. Interest in cobalt compounds was renewed by Mushett et al. (1952), who demonstrated in 1952 that hydroxocobalamin (vitaminB12).Paulet (1960) subsequently reported that cobalt EDTA was moreeffective as a cyanide antidote than the classic nitrite-thiosulfatecombination.Hydrogen cyanide is used in the fumigation of ships, largebuildings, flour mills, private dwellings, freight cars, andbound to a carrier, commonly diatomaceous earth, and blended with anaeroplanes that have been infested by rodents or insects. It is odorous or irritating product as a warning marker.Halogenated cyanides (chloro-, bromo- and iodocyanide) inCyanide salts are utilized in metal cleaning, hardening, ore-extracting processes, and electroplating. contact with water produce the non-toxic cyanic acid. As a resultis used as a solvent and is less toxic (LD50 = 120 mg/kg) thanof contact with strong acids, hydrogen cyanide is liberated. Nitriles are cyano-derivatives of organic compounds. Acetonitrilehydrogen cyanide (LD50= 0.5 mg/kg), but often contains toxicadmixtures due to metabolism to inorganic cyanide. While aliphatic nitrilesmetabolise to inorganic cyanide, the aromatic nitrile bond is stablein vivo. Acrylonitrile is the raw material used for themanufacture of plastics and synthetic fibres. Contact with skincauses bullae formation. Pyrolysis generates hydrogen cyanide.Acrylonitrile and propionitrile are less toxic (LD50 = 35 mg/kg)than butyronitrile (LD50 = 10 mg/kg). Trichloroacetonitrile(LD50 = 200 mg/kg) is used as an insecticide. The aromaticnitriles, bromoxynil (LD50= 190 mg/kg) and ioxynil (LD50= 110mg/kg), are used as herbicides.Cyanamide, cyanoacetic acid, ferricyanide and ferrocyanide donot release cyanide. They are therefore less toxic (LD50=1000-2000 mg/kg) than the cyanogenic compounds above, though theymay cause toxicity by other means, e.g. cyanide in combination withalcohol.Fires and automobile pollution-control devices withmalfunctioning catalytic converters (Voorhoeve et al., 1975)generate cyanide. Natural substances, such as wool, silk, horsehair, and tobacco, as well as modern synthetic materials, such aspolyurethane and polyacrylonitriles, release cyanide duringHarland, 1982; Clark et al., 1983; Alarie, 1985; Lowry et al., 1985)combustion (Levine et al., 1978; Birky et al., 1979; Anderson & (Table 1).Table 1. Hydrogen cyanide generated by pyrolysisµg HCN perMaterial g materialpaper 1100wool 6300cotton 130 nylon 780polyurethane foam 1200From: Montgomery et al. (1975)Cyanide is found in foodstuffs such as cabbage, spinach, andalmonds, and as amygdalin in apple pips, peach, plum, cherry, andcompletely harmless as long as it is relatively dry. However, thealmond kernels. In the kernels themselves, amygdalin seems to beC20H27NO11 + 2H2O --> 2C6H12O6 + C6H5CHO + HCNseeds contain an enzyme that is capable of catalysing the following hydrolytic reaction when the seeds are crushed and moistened:amygdalin glucose benzaldehyde hydrogencyanideNatural oil of bitter almonds contains 4% HCN. American whiteThe reaction is slow in acid but rapid in alkaline solution. lima beans contain 10 mg cyanide/100 g bean. The dried root ofto be about 9 mg and that in wild apricot seeds more than 200 mg.cassava (tapioca) may contain 245 mg cyanide/100 g root. The cyanide content in 100 g of cultivated apricot seeds has been foundCyanide is also formed during nitroprusside therapy, especiallywhen it is prolonged, because tachyphylaxis sometimes requires the(Smith & Kruszyna, 1974; MacRae & Owen, 1974; Piper, 1975; Atkins,use of higher doses than the recommended maximum of 10 µg/kg per minantihypertensive agents and they saw wide use because they were very1977; Anon, 1978). Cyanide metabolises to thiocyanate. Thiocyanates were used some years ago asled to their disfavour.effective. However, a variety of subacute toxic effects, including anorexia, fatigue, and gastrointestinal tract and CNS disturbances,effect could not be demonstrated in either retrospective orLaetrile, amygdalin derived from apricot kernels, has been used as an anticancer agent, but it is now obsolete because a therapeutic prospective studies. Laetrile has caused fatal cyanide poisoning (Sadoff et al., 1978).It is generally accepted that inhalation of approximately 50 ml(at 1.85 mmol/l) of hydrogen cyanide gas is fatal within minutes.accidental than suicidal. Thus accidental cyanide poisoning mayPoisoning from hydrogen cyanide is more frequentlycourse of their work (Chen et al., 1944). In fires, a combinationoccur in fumigators and chemists who use hydrogen cyanide during theSuicidal ingestion of cyanide salts most commonly occurs inof HCN and carbon monoxide (CO) toxicity, as a result of inhalation of combustion products, may cause fatalities.et al., 1982). However, death may be delayed for several hourspersonnel with occupational access to cyanide. The ingestion of as little as 250 mg of an inorganic cyanide salt may be fatal (Peters following the ingestion of cyanide on a full stomach; a first-pass effect in the liver may also delay the onset of toxicity (Naughton, 1974).Chronic low-dose neurotoxicity have been suggested byepidemiological studies of populations ingesting naturally occurringplant glycosides (Blanc et al, 1985). These glycosides are presenta major tropical foodstuff (Conn, 1973; Cook & Coursey, 1981;in a wide variety of plant species, most notably the cassava plant, Ministry of Health, Mozambique, 1984). Cassava has been associatedcyanide and a low sulfur intake from diets dominated bywith tropical ataxic neuropathy (Cook & Coursey, 1981). Epidemic spastic paraparesis has been associated with a combination of a high insufficiently processed cassava and lacking protein supplementaryal., 1983). Long-term cyanide intoxication has been shown to befood (Rosling, 1989). A neurotoxicological role for cyanide has also been suggested in tobacco-associated amblyopia (Grant, 1980) and in amygdalin-associated peripheral neuropathy (Kalyanaraman et associated both with thyroid gland enlargement and dysfunction in case reports and in cohort studies of individuals exposed occupationally (Blanc et al., 1985), through dietary intake (Cook & Coursey, 1981), and experimentally (El Ghawabi et al., 1975).Cyanide has a special affinity for the ferric ions that occurin cytochrome oxidase, the terminal oxidative respiratory enzyme inutilization of oxygen. When cytochrome oxidase is inhibited bymitochondria. This enzyme is an essential catalyst for tissue cyanide, histotoxic anoxia occurs as aerobic metabolism becomesrelease of biogenic amines may play a role by causing cardiacinhibited. In massive cyanide poisoning, the mechanism of toxicity is more complex. It is possible that autonomic shock from thedecrease in cardiac output. This theory is supported by the sharpfailure (Burrows & Way, 1976). Cyanide could cause both pulmonary arteriolar and/or coronary arterial vasoconstriction, which would result, either directly or indirectly, in pump failure and aobservation that phenoxybenzamine, an alpha-adrenergic blockingincrease in central venous pressure that was observed by Vick & Froelich (1985) at a time when the arterial blood pressure fell after the intravenous administration of sodium cyanide to dogs. Thenitrite, a potent arteriolar vasodilating agent, resulted in thedrug, partially prevented these early changes (Vick & Froelich, 1985) supports the concept of an early shock-like state not related to inhibition of the cytochrome oxidase system. Inhalation of amyl survival of dogs in these experimental circumstances. This could have been due to reversal of early cyanide-induced vasoconstriction with restoration of normal cardiac function (Vick & Froelich, 1985).The smell of bitter almonds in expired air is an important signin cyanide poisoning. However, many people are unable to perceiveincidence of "non-smellers" is reported to be 18% among males and 5%the odour of hydrocyanic acid (Kalmus & Hubbard, 1960). The among females (Kirk & Stenhouse, 1953; Fukumoto et al., 1957).experienced. A blood-stained aspirate may be observed if gastricImmediately after swallowing cyanide, very early symptoms, such as irritation of the tongue and mucous membranes, may be lavage is performed. Early symptoms and signs that occur aftercyanosis, hypotension, bradycardia, and sinus or AV nodalinhalation of HCN or the ingestion of cyanide salts include anxiety, headache, vertigo, confusion, and hyperpnoea, followed by dyspnoea, arrythmias. In the secondary stage of poisoning, impaired consciousness,hypotension, complex arrythmias, cardiovascular collapse, pulmonarycoma and convulsions occur and the skin becomes cold, clammy, and moist. The pulse becomes weaker and more rapid. Opisthotonos and trismus may be observed. Late signs of cyanide toxicity include oedema, and death.the high concentration of oxyhaemoglobin in the venous return, but,It should be emphasized that the bright-red coloration of the skin or absence of cyanosis mentioned in textbooks (Gosselin et al., 1984; Goldfrank et al., 1984) is seldom described in case reports of cyanide poisonings. Theoretically this sign could be explained bydifferent mechanisms: (1) an intracellular metabolic process thatespecially in massive poisoning, cardiovascular collapse will prevent this from occurring. Sometimes, cyanosis can be observed initially, while later the patient may become bright pink (Hilmann et al., 1974). The pathogenesis of pulmonary oedema could be due to severalpoisoning and it has been shown that cyanide significantly increasescould injure the alveolar and capillary epithelium directly, producing a capillary leak syndrome; (2) neurogenic pulmonary oedema or, (3) most likely, a direct effect on the myocardium leading to left ventricular failure and increased pulmonary venous pressure. The brain is obviously the key organ involved in cyanide brain lactate and decreases brain ATP concentrations (Olsen & Klein, 1947).Since oxidative phosphorylation is blocked, the rate ofglycolysis is markedly increased, which in turn leads to lacticacidosis. The degree of lactic acidosis can be correlated with theseverity of cyanide poisoning (Trapp, 1970; Naughton, 1974).A reversible toxic effect occurs on the pancreatic beta-cells,which may occasionally give rise to an erroneous diagnosis of hyperglycaemic diabetic coma.Before intravenous treatment with antidotes is commenced, it isnecessary to collect a heparinized (not fluoride) blood sample forcollected after treatment are totally unreliable. A quantitativedetermination of cyanide concentration. Results from samples test employing a detector tube (see chapter 10) can be used if thepoisoning can be evaluated. Therapeutic measures after antidotaldiagnosis is in doubt. The blood can also be used for a quantitative test (see chapter 10), so that the severity of treatment should be based on the clinical condition of the patientto 0.005-0.04 mg/l have been recorded in healthy non-smokers, andrather than on blood cyanide concentrations (Berlin, 1971; Vogel et al., 1981; Peters et al., 1982). Since blood concentrations of up 0.01-0.09 mg/l in smokers, only concentrations above these values(plasma), 6.0 µg/l (erythrocytes); smokers 8.6 µg/l (whole blood),were previously considered to be toxic (Vogel et al., 1981; Peters et al., 1982). Lundquist et al., (1985) reported even lower concentration: non-smokers 3.4 µg/l (whole blood), 0.5 µg/l 0.8 µg/l (plasma), 17.7 µg/l (erythrocytes).bound. A plasma-to-blood ratio as high as 1:10 has been reportedFatal cyanide poisoning has been reported with whole blood concentrations of >3 mg/l and severe poisoning with 2 mg/l (Graham et al., 1977). However, when cyanide enters the bloodstream, up to 98% quickly enters the red blood cells where it becomes tightly1976). However, a serious drawback to the use of plasma cyanideand, as a consequence, the whole blood cyanide concentration may not accurately reflect tissue concentrations of cyanide. Plasma levels of cyanide may be of greater significance because severe toxicity occurs in the presence of only modest concentrations (Vesey et al., determinations in the assessment of poisoning is the pronounced instability of cyanide in plasma (Lundquist et al., 1985).The major pathway of endogenous detoxification is conversion,by means of thiosulfate, to thiocyanate. Minor routes of eliminationare excretion of hydrogen cyanide through the lungs and binding.Metabolic Detoxification of Cyanide;V02ANnew.BMPto cysteine or hydroxocobalamin. The detoxification of cyanide occurs slowly at the rate ofenzyme is needed to catalyse the transfer of a sulfur atom0.017 mg/kg per min (McNamara, 1976). A sulfurtransferase from the donor thiosulfate to cyanide. The classical theorydoubt because thiosulfate penetrates lipid membranes slowlyindicating that mitochondrial thiosulfate sulfurtransferase is the most important enzyme in this reaction is now in and would, therefore, not be readily available as aprimary cyanide detoxification buffer operating in normal metabolismsource of sulfur in cyanide poisoning. The modern concept assumes a greater role for the serum albumin-sulfane complex, which is the (Sylvester et al., 1983). A further enzyme, beta-mercaptopyruvate sulfurtransferase, also converts cyanide to thiocyanate (Vesey et al., 1974). This enzyme is found in the erythrocytes, but in human cells its activity is low.The detoxification product of cyanide, thiocyanate, is excretedin the urine. Thiocyanate concentrations are normally betweenplasma half-life of thiocyanate in patients with normal renal1-4 mg/l in the plasma of non-smokers and 3-12 mg/l in smokers. Thetherefore at increased risk of toxicity (Schulz et al., 1978).function is 4 h (Blaschle & Melmon, 1980), but in those with renal insufficiency it is markedly prolonged and these patients aremuscle spasm, nausea, disorientation, psychosis, hyper-reflexia, andThiocyanate levels exceeding 100 mg/l are thought to be associated with toxicity. Thiocyanate toxicity is characterized by weakness,1931; Garvin, 1939; Russel & Stahl, 1942; Kessler & Hines, 1948;stupor (Smith, 1973; Michenfelder & Tinker, 1977). Lethal poisoning at concentrations greater than 180 mg/l has been reported (Healy, Domalski et al., 1953). Haemodialysis is recommended as anDialysance values of 82.8 ml/min ( in vivo) and 102.3 ml/mineffective means of removing thiocyanate (Marbury et al., 1982).( in vitro) have been recorded (Pahl & Vaziri, 1982). Little isknown about the protein-binding characteristics of thiocyanate, andhaemoperfusion may be more effective than haemodialysis.Accidental exposure to cyanide, as either hydrogen cyanide orcyanide salts, will occur primarily in the occupational context, andappropriate preventive and protective measures need to be takenwherever cyanides are manufactured or used. Many industrial accidentsoccur as a result of mixing cyanide salts and acids, and care should be taken when both are present on industrial premises.The public may be affected in the case of a major industrialAs hydrogen cyanide may be generated during combustion of organic substances, fire fighters may also be exposed occupationally.cyanides are used to have contingency plans that will enable them toemergency, or of a transport accident, involving the release of cyanides. It is essential for local authorities in areas whereindustrial hygiene are essential for the prevention of cyaniderespond effectively. Adequate hospital facilities for treatment of casualties must be available. Proper maintenance of plant, good operating practice, andpeople in zones where cyanide could be released accidentally. Therepoisoning. Areas in the workplace where cyanides are used and containers for storage and transport of cyanide should be clearly marked. Work schedules should ensure that there are at least twotoxic materials, such as dirty and clean locker facilities andshould be showers and first-aid kits in these areas. Personnel without proper training should not be allowed in the plant. Normal industrial and laboratory hygiene measures for personnel handlingcyanides, should receive instruction on the dangers of cyanides andshowers, should be provided. Eating, drinking, and smoking should not be allowed in the work area where cyanides are used but in places specially reserved for these purposes. Each employee working at a plant or laboratory that handlessymptoms and signs of cyanide poisoning and how to achieve safebe trained in appropriate first-aid measures, as should emergency-service personnel. They should be aware of the hazards and informed about the possible routes of exposure (inhalation, skin absorption, ingestion). Training should involve recognition of theregular instruction sessions covering procedures for handlingremoval of victims from the source of intoxication. Personnel should also be able to guide a rescue or fire-fighting team to a trapped intoxicated person. Rescue personnel should be able to put on protective clothing quickly in an emergency. There should beand decontamination of exposed skin and eyes. It should be realizedcyanides and for rescue in case of accidents, as well as random alarm exercises. First-aid training should include the essential measures to be taken before medical help arrives, which may need to be undertaken at the same time as removal of contaminated clothingin plants where the gas is used or may be generated. Warningthat further uptake of cyanide into the blood may occur after showering because of continued skin absorption. Each plant handling cyanide should have its own medical staff trained in the emergency treatment of cyanide poisonings. The atmospheric concentrations of hydrogen cyanide should be monitoredFilter respirators should be carried at all times by employeesdevices are available for this purpose and should be installed. In certain circumstances in which cyanide is used, it is possible to add a warning gas, e.g., cyanogen chloride and chloropicrin have been added to hydrogen cyanide used as a fumigant (Cousineau & Legg, 1935; Polson & Tattersall, 1969). working in zones where hydrogen cyanide may be released. At highworker should be aware of the emergency procedures to be followedhydrogen cyanide concentrations, absorption occurs through the skin and impermeable butyl rubber protective clothing is required. Oxygen breathing apparatus may be needed. In the case of an accident involving hydrogen cyanide there should be both an acoustic and a visual alarm for the plant, which may be activated by workers in zones where the gas is used. Eachhave the authority and training to perform the special resuscitationand the protective clothing and equipment to be used. If a large number of victims is involved or if there is a danger to the public, local authorities need to be warned, so that contingency plans are put into effect and hospitals alerted. For accidents at plants in remote areas where a qualified physician is not readily available and there are no hospital intensive care facilities, attending paramedical personnel should measures involved in treating cyanide poisonings, including rapid endotracheal intubation and techniques for obtaining intravenous access.Although effective antidotes are available, general supportivemeasures should not be ignored and may be life-saving.experience of 104 industrial poisoning cases, the use of specificAccording to Jacobs (1984), who reported his personaldeep coma, wide non-reactive pupils, and respiratory insufficiencyantidotes was indicated only in cases of severe intoxication withmoderately severe poisoning, who had suffered only a brief period ofin combination with circulatory insufficiency. In patients with unconsciousness, convulsions, vomiting, and cyanosis, therapycases of mild intoxication with dizziness, nausea, and drowsiness,consisted of intensive care and intravenous sodium thiosulfate. In rest and oxygen alone were used. Peden et al. (1986) described nine patients poisoned byfrom the area where they had been working. The arterial whole-bloodhydrogen cyanide released by a leak from a valve. Three of them were briefly unconscious but recovered rapidly after being moved cyanide concentrations on admission were 3.5, 3.1 and 2.8 mg/l,two were transiently unconscious, and in these cases the cyaniderespectively. The cyanide concentrations in the other cases ranged between 2.6 and 0.93 mg/l. All recovered with supportive therapy alone. Between 1970 and 1984, three other men were treated similarly;comatose patients with potentially lethal blood concentrations onconcentrations 30 min after exposure were 7.7 and 4.7 mg/l. The concentration in the other patient was 1.6 mg/l. All three patients recovered without the use of cyanide antidotes. Small numbers of admission, and who recovered without cyanide antidotes, have beenA patient exposed to hydrogen cyanide who reaches hospitalreported by Graham et al. (1977), Edwards & Thomas (1978), and Vogel et al. (1981). Even if a patient is unconscious, an antidote does not necessarily have to be administered immediately unless vital signs deteriorate. fully conscious is only likely to require observation and reassurance. 1.9.2.1 OxygenIt is difficult to understand how oxygen has a favourableeffect in cyanide poisoning, because inhibition of cytochromeregarded as an important first-aid measure in cyanide poisoning, andoxidase is non-competitive. However, oxygen has always been there is now experimental evidence that oxygen has specificinhibition by cyanide (Takano et al., 1980). Nevertheless, thereantidotal activity. Oxygen accelerates the reactivation of cytochrome oxidase and protects against cytochrome oxidasesuffering from combined carbon monoxide and cyanide poisoning, sinceare other possible modes of action and those that are clinically important have yet to be determined. Hyperbaric oxygen is recommended for smoke inhalation victimsThe major route of cyanide detoxification in the body isthese two agents are synergistically toxic. The use of hyperbaric oxygen in pure cyanide poisoning remains controversial. 1.9.2.2 Sodium thiosulfate conversion to thiocyanate by rhodanese, although otherCyanide poisoning is an intramitochondrial process and ansulfurtransferases, such as beta-mercaptopyruvate sulfurtransferase, may also be involved. This reaction requires a source of sulfane sulfur, but endogenous supplies of this substance are limited. intravenous supply of sulfur will only penetrate mitochondriafor example in cases of smoke inhalation. Sodium thiosulfate isslowly. While sodium thiosulfate may be sufficient alone in mild to moderately severe cases, it should be administered with other antidotes in cases of severe poisoning. It is also the antidote of choice when the diagnosis of cyanide intoxication is not certain,for many years as a simple first-aid measure that generatesassumed to be intrinsically nontoxic but the detoxification product formed from cyanide, thiocyanate, may cause toxicity in patients with renal insufficiency (see section 1.7). 1.9.2.3 Amyl nitrite The administration of amyl nitrite by inhalation has been usedrecent studies suggest that methaemoglobin formation plays only amethaemoglobin and which can be employed by lay personnel. Its use was abandoned because the methaemoglobin concentration obtained with amyl nitrite is no more than 7% and it is thought that at least 15% is required to bind a potentially lethal dose of cyanide. However, small role in the therapeutic effect of amyl nitrite, andNitrites generate methaemoglobin, which combines with cyanidevasodilatation may be the most important mechanism of antidotal action. Artificial respiration with amyl nitrite ampoules broken into an Ambu bag proved to be life-saving in dogs severely poisoned with cyanide. Amyl nitrite may therefore be reintroduced as a first-aid measure. 1.9.2.4 Sodium nitrite to form the nontoxic substance cyanmethaemoglobin. Methaemoglobinhaemoglobin should be monitored to ensure aerobic metabolism of thedoes not have a higher affinity for cyanide than does cytochrome oxidase, but there is a much larger potential source of methaemoglobin than there is of cytochrome oxidase. The efficacy of methaemoglobin is therefore primarily the result of mass action. A drawback of methaemoglobin generation is the resultant impairment of oxygen transport to cells and, ideally, the total amount of freelikely to be less because only low plasma concentrations arecells. Methaemoglobin can be measured very quickly, but this in itself will not provide an accurate guide to the amount of haemoglobin available for oxygen transport because the cyanmethaemoglobin concentration is not taken into account. Individuals deficient in glucose-6-phosphate dehydrogenase (G6PD) are at great risk from sodium nitrite therapy because of the likelihood of severe haemolysis, but the risk from amyl nitrite isassociated with methaemoglobin formation, as described above forachieved. Excess methaemoglobinaemia may be corrected with either methylene or toluidine blue (see Chapter 9) or, preferably, where feasible, by exchange transfusion. 1.9.2.5 4-Dimethylaminophenol (4-DMAP) 4-DMAP generates a methaemoglobin concentration of 30-50% within a few minutes (Weger, 1968) and, theoretically, it should therefore be valuable as a first-aid measure. However, the problems nitrites, apply to 4-DMAP to an even greater extent. Furthermore,1.9.2.6 Hydroxocobalamin (vitamin Bl2a)it has very poor dose-response curve reproducibility. Haemolysis as a result of 4-DMAP therapy has been observed in overdose as well as following a correct therapeutic dose. Treatment with 4-DMAP is contraindicated in patients with G6PD deficiency. Excess methaemoglobinaemia may be corrected with either methylene or toluidine blue (see section 1.9.2.8).This antidote binds cyanide strongly to form cyanocobalamin(vitamin B12) and, compared to nitrite and 4-DMAP therapy, it hasthe great advantage of not interfering with tissue oxygenation. Thedisadvantage of hydroxocobalamin as a cyanide antidote is the largecyanide (corresponding to 65 mg KCN) needs 1406 mg hydroxocobalamin.dose required for it to be effective. Detoxification of 1 mmol In most countries it is only commercially available in formulationsthat has to be reconstituted with 80 ml of a 10% sodium thiosulfateof 1-2 mg per ampoule. In some countries, e.g., France, a formulation is available that contains 4 g hydroxocobalamin powderreactions and acne. Some authors have reported a reduced antidotalsolution prior to use and administered intravenously in a minimum of 220 ml of 5% dextrose. Recorded side effects are anaphylactoidHistological changes in the liver, myocardium, and kidney apparentlyeffect as a result of mixing hydroxocobalamin and sodium thiosulfate in the same solution (Evans, 1964; Friedberg & Shukla, 1975). induced by hydroxocobalamin have been reported in animal1.9.2.7 Dicobalt edetateexperiments (Hoebel et al., 1980), but their relevance to man has not yet been established. Transient pink discoloration of mucous membranes and urine is an unimportant and nontoxic side-effect.definitely present. This is a strict criterion, because as a resultThis agent has been shown to be effective in the treatment of cyanide poisoning in man, and in the United Kingdom it is the current treatment of choice provided that cyanide toxicity is of the manufacturing process some free cobalt ions are alwaysthat glucose protects against cobalt toxicity and it is recommendedpresent in solutions of dicobalt edetate. Cobalt ions are toxic and the use of dicobalt edetate, in the absence of cyanide, will lead to serious cobalt toxicity. There is evidence from animal experiments that this be given at the same time as dicobalt edetate. Seriousconcentrations in the presence of cyanide is difficult.adverse effects recorded include vomiting, urticaria, anaphylactic shock, hypotension, and ventricular arrhythmias (Hilmann et al., 1974; Naughton, 1974). 1.9.2.8 Antidotes to methaemoglobin-forming agents Accurate determination of methaemoglobin and free haemoglobin Nevertheless, excess methaemoglobinaemia does undoubtedly occur on occasions following the use of nitrites and 4-DMAP. Excess methaemoglobin concentrations may be reduced by methylene or toluidine blue. However, regeneration of haemoglobin will release cyanide back into the circulation, leading to a recurrence of toxicity.The management of cyanide poisoning is determined by (i) thenature of exposure, i.e. hydrogen cyanide (with or without carbonmonoxide), cyanide salts, aliphatic nitriles, cyanogenic glycosides;(ii) the severity of poisoning; (iii) the number of patientsinvolved; (iv) the proximity of hospital facilities;
Sumber :Internet
Tidak ada komentar:
Posting Komentar